Aluminum
What is Aluminum
Aluminum (pronounced as ah-LOO-men-em) is a soft and lightweight metal, represented by the chemical Al. It exists in the form of several mineral compounds, having a wide range of commercial uses due to its non-toxic nature and high-corrosion resistance. It has 15 isotopes with mass numbers ranging from 22 to 35 of which only Al-26 is radioactive with a half life of 7.17×105years and Al-27 exists naturally.
Where Can it Be Found
The element constitutes about 8.1% of the earth’s crust making it one of the most abundant metals. It can be found combined with other metals in the form of silicate minerals like cryolite and bauxite. Its production on a commercial scale is done by an expensive chemical process called Hall-Heroult process that involves a large amount of energy.
History
Origin of its Name: Its name is derived from the Latin word for alum called ‘alumen’ that means bitter salt [1].
Who Discovered it: Hans Oersted [1]
When, Where, And How was it Discovered
It was believed during the 3rd century in China that the tomb of a military leader called Chou-Chu contained about 85% aluminum. In the 1700s, the presence of the metal’s oxide was also coined, but no further analysis could be made. In fact, the Cornish chemist Henry Davy tried to extract sodium and potassium from their respective oxides, but the Al could not be obtained.
It was only in 1825 when Oersted at Copenhagen, Denmark heated aluminum chloride with potassium and extracted an impure form of the metal. In 1827 Friedrich Wohler, the German chemist, finally repeated the method using sodium and obtained a pure sample [1].
Classification, Properties, and Characteristics of Aluminum
General Properties | |
| Relative/Average atomic mass | 26.982 [1] |
| Atomic mass/weight | 26.982 atomic mass units [4] |
| Molar mass/Molecular weight | 26.982 g/mol [3] |
| Mass number | 27 |
Physical Properties | |
| Color/physical appearance | Silver white[1] |
| Melting point/freezing point | 660.323°C (1220.581°F) [1] |
| Boiling point | 2519°C (4566°F) [1] |
| Density | 2.70 g/cm3 [1] |
| Standard/Physical state of matter at normal room temperature (solid/liquid/gas) | Solid [1] |
| Ductility | Yes |
| Malleability | Yes |
| Hardness | 2.9 Mohs [6] |
| Electrical conductivity | 37.6676 x 106 Sm-1[4] |
| Thermal conductivity | 237 Wm-1K-1 [4] |
| Specific heat capacity | 0.90 Jg-1K-1 [4] |
| Specific gravity | 2.8 |
| Resistivity | 2.65 x10-8ρ (ohm m) [5] |
| Tensile strength | 90 Mpa [5] |
| Temperature coefficient | 0.00393 α/°C |
| Tensile yield strength | 276 Mpa [7] |
| Coefficient of linear thermal expansion/heat transfer | 23.1 10-6K-1 |
| Coefficient of friction | 1.35 |
Chemical Properties | |
| Flammability | Flammable [4] |
| Oxidation state/Oxidation number | +1, (+2), +3 [1] |
Atomic Data of Aluminum
| Valence electrons | 1 [8] | ||||||
| Electron configuration (noble gas configuration) | [Ne]3s23p1[1] | ||||||
| Crystal structure | Face-centered cubic | ||||||
| Lattice constant/parameter | 0.405 nm | ||||||
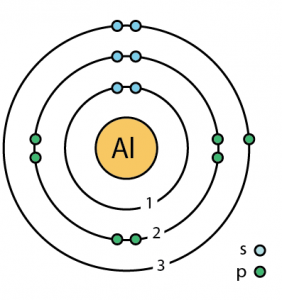
| Energy levels [9] | |||||||
| First energy level | 2 | ||||||
| Second energy level | 8 | ||||||
| Third energy level | 3 | ||||||
| Atomic structure [4] | |||||||
| – Number of electrons | 13 | ||||||
| – Number of neutrons | 14 | ||||||
| – Number of protons | 13 | ||||||
| Radius of atom | |||||||
| – Atomic radius | 1.84 Å [1] | ||||||
| – Covalent radius | 1.24 Å [1] | ||||||
| Electronegativity | 1.61 [1] | ||||||
| Ion charge | +1 [3] | ||||||
| Ionization energy [1]
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 577.539 | 1816.679 | 2744.781 | 11577.469 | 14841.857 | 18379.49 | 23326.3 | |
What is Aluminum Commonly Used for
- It is used in the a vast range of products like fence gate panels, sidings, cans, tubing, pipes, wirings, foils, roofing, frying, pans, railings, coatings, patio covers, wheels, brazing rods, screen doors, radiators, gas fuel tanks, and poles [1].
- A good conductor of electricity, Al makes an ideal substitute for copper in terms of cost and weight to make electrical transmission lines [1].
- Aluminum is alloyed with other metals like copper, magnesium, silicon, and manganese to make ball bearings, automobile parts, rockets, and airplanes [1, 2].
- The metal is often used as a reflective coating on telescope mirrors, toys, packages, decorative, and decorative papers to protect against light and heat [1].
- As an alternative to expensive batteries and liquid fuels, aluminium fuel ion air cells/batteries are more convenient and portable for use [10].
Does the Element Have Any Toxic Effects
Inhalation of Al-containing dusts or fumes affects the respiratory system, causing cough and other lung-related disorders. Prolonged ingestion of the metal in the form of some oral products can be damaging to the kidneys as well as increase the possibility of Alzheimer’s disease [11].
Interesting Facts
- Before the discovery of the Hall-Heroult process, pure Al was more expensive than gold and hence called the ‘King of Metals’ [12].
- The metal can be recycled several times without compromising on its quality [12].
Aluminum Price
The cost of pure may vary somewhere between $ 0.9 and $ 1.
References
- http://www.rsc.org/periodic-table/element/13/aluminium
- https://education.jlab.org/itselemental/ele013.html
- https://pubchem.ncbi.nlm.nih.gov/compound/aluminum#section=Computed-Properties
- https://www.chemicool.com/elements/aluminum.html
- http://hyperphysics.phy-astr.gsu.edu/hbase/Tables/rstiv.html
- Lbma.org.uk
- http://asm.matweb.com/search/SpecificMaterial.asp?bassnum=ma6061t6
- https://study.com/academy/answer/how-many-valence-electrons-does-aluminum-have.html
- http://www.chemicalelements.com/elements/al.html
- https://www.sciencedirect.com/science/article/pii/S0378775307002236
- https://www.atsdr.cdc.gov/phs/phs.asp?id=1076&tid=34