Chlorine
What is Chlorine
Chlorine (pronounced as KLOHR-een) is a highly reactive diatomic gas belonging to the family of halogens. Denoted by the chemical symbol Cl, it categorized as a nonmetal [1, 2]. It has 16 isotopes with mass numbers ranging from 31 to 46 of which only Cl-35 and Cl-37 have a natural existence with a stable half-life [3].
Where is the Element Found
It does not occur freely in nature, existing in minerals like halite, carnallite, and sylvite. It is also found in the form of sodium chloride in salt beds and lakes that have occurred as a result of leaching into the sea since ages [1].
History
Origin of its Name: Its name is derived from the Greek word ‘chloros’ that means greenish yellow [1].
Who Discovered it: Carl Wilhelm Scheele
Discovery Date: 1774
When, Where, and How was Chlorine Discovered
In 1774 at Uppsala, Sweden, when Scheele heated hydrochloric acid with the mineral pyrolusite, a greenish-yellow gas was produced that had a choking odor. It also dissolved in water to produce an acidic solution, and bleached litmus paper, flowers, and leaves.
In 1807, the Cornish chemist Humphry Davy found the new gas to be an element, officially announcing its existence in 1807. However, it took another ten years for chlorine to be recognized as an element by other chemists [1, 2].
Identification |
|||
| Atomic number | 17 [1] | ||
| CAS number | 7782-50-5 [1] | ||
| Position in the periodic table [1] | Group | Period | Block |
| 17 | 3 | p | |
Classification, Properties, and Characteristics of Chlorine
General Properties |
||
| Relative/Average atomic mass | 35.45 [1] | |
| Atomic mass/weight | 35.45 atomic mass units [5] | |
| Molar mass/Molecular weight | 70.9 g/mol [6] | |
| Mass number | 35 | |
Physical Properties |
||
| Color/physical appearance | Yellowish green [1] | |
| Odor | Pungent [1] | |
| Melting point/freezing point | -101.5°C (-150.7°F) [1] | |
| Boiling point | -34.04°C (-29.27°F) [1] | |
| Density | 0.002898 g/cm3 [1] | |
| Standard/Physical state of matter at normal room temperature (solid/liquid/gas) | Gas [1] | |
| Electrical conductivity | Unknown [4] | |
| Thermal conductivity | 0.0089 Wm-1K-1 [4] | |
Chemical Properties |
||
| Flammability | Non-flammable [1] | |
| Oxidation state/Oxidation number | -1, +1, +2, +3, +4, +5, +6, +7 [1] | |
Atomic Data of Chlorine (Element 17)
| Valence electrons | 7 [7] | ||||||
| Electron configuration (noble gas configuration) | [Ne] 3s23p5[1] | ||||||
| Atomic structure [3] | |||||||
| – Number of Electrons | 17 | ||||||
| – Number of Neutrons | 18 | ||||||
| – Number of Protons | 17 | ||||||
| Radius of atom | |||||||
| – Atomic radius | 1.75 Å [1] | ||||||
| – Covalent radius | 1.00 Å [1] | ||||||
| Electronegativity | 3.16 [10] | ||||||
| Ion charge | -1 | ||||||
| Ionization energy [1]
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 1251.186 | 2297.663 | 3821.78 | 5158.608 | 6541.7 | 9361.97 | 33603.91 | |
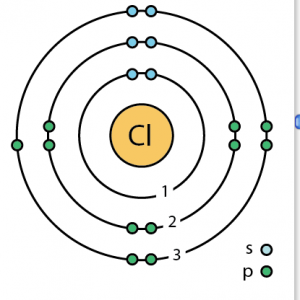
Chlorine Atomic Structure (Bohr Model)
What are the Common Uses of Chlorine Gas
- Chlorine-based liquids, granules, and tablets are used as disinfectants to treat swimming pools and drinking water [1].
- It is added to PVC (polyvinyl chloride) used in electrical wiring insulation, window frames, water pipes, blood bags, and car interiors [1].
- Commercial household bleaches containing Cl are effective in cleaning kitchen and bathroom areas, and whitening clothes [3].
- The element is used in the manufacture of several consumer products like paints, dyes, solvents, paper, insecticides, and textiles [1].
- Some medicines and pharmaceutical products used in the treatment of arthritis, allergy, and high cholesterol contain chlorine as the active ingredient [1, 3].
- It finds application in certain oxidation and substitution reactions [1].
What are the Toxic Effects of Chlorine
Prolonged exposure to chlorine gas can cause watery eyes, skin rash, burns, shortness of breath, nausea, and vomiting. Inhalation of high concentration of the gas can lead to severe health hazards inhalation like pulmonary edema [8].
Interesting Facts
It was used as a chemical weapon in the World War I by the Germans for its toxic nature [1].
Salt water can be electrolyzed to release chlorine [1].
Chlorine Cost
The price of pure chlorine may vary between $0.15 and $0.16.
References
- http://www.rsc.org/periodic-table/element/17/chlorine
- https://education.jlab.org/itselemental/ele017.html
- https://www.chemicalsafetyfacts.org/chlorine/
- https://www.chemicool.com/elements/chlorine.html
- https://hobart.k12.in.us/ksms/PeriodicTable/chlorine.htm
- https://pubchem.ncbi.nlm.nih.gov/compound/chlorine#section=Top
- https://www.toppr.com/ask/content/story/anomalous-properties-of-fluorine-75571/
- https://emergency.cdc.gov/agent/chlorine/basics/facts.asp