Copernicium
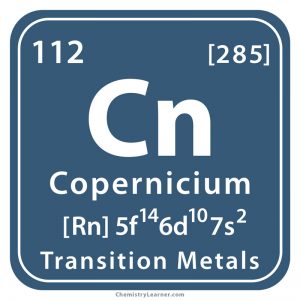
Copernicium (pronounced as kogh-per-NEE-see-em), previously known as ununbium, is a synthetic element, denoted by the symbol Cn [1, 3].
History
Who discovered it
On February 9, 1996, German scientists Sigurd Hofmann, Peter Armbruster, Gottfried Münzenbe, and Bulgarian researcher Victor Ninov produced copernicium at the GSI Helmholtz Centre for Heavy Ion Research in Darmstadt, Germany. Copernicium- 277, an isotope with a half-life of 0.24 milliseconds, was synthesized by bombarding Lead-208 with Zinc-70 using a linear accelerator.
Although several attempts were made to generate other isotopes, isotope-285, a decay product of flerovium, was found to be the most stable with a half-life of 30 seconds in a research at the Joint Institute for Nuclear Research (JINR), Dubna, Russia [1, 2, 3].
How did Copernicium get its name
It is named after renowned mathematician and astronomer Nicolaus Copernicus who originated the solar system model that led to the introduction of the Bohr model of the atom [1, 8].
Position of Copernicium in the Periodic Table [1]
| Group | 12 |
| Period | 7 |
| Block | d |
Copernicium Physical Properties [1]
| Relative Atomic Mass | 285 |
| Color/Appearance | Unknown |
| Odor | Unknown |
| Boiling Point | Unknown |
| Melting Point | Unknown |
| Density | Unknown |
| State at Room Temperature | Solid (unconfirmed) |
| Malleability | Unknown |
Copernicium Chemical Characteristics [1]
| Flammability | Unknown |
| Radioactivity | Highly radioactive |
| Oxidation state/Oxidation number | [+2], [+4] |
Atomic Data of Copernicium [1, 6]
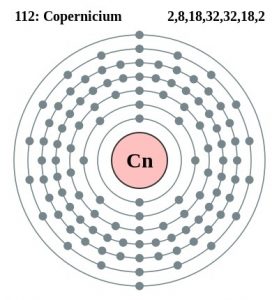
| Atomic Number | 112 |
| Electron Configuration | [Rn] 5f14 6d10 7s2 |
| Crystal Structure | Hexagonal close packed |
| Atomic Structure | |
| – Number of Electrons | 112 |
| – Number of Neutrons | 277 |
| – Number of Protons | 112 |
| Radius of Atom | |
| – Atomic Radius | Unknown |
| – Covalent Radius | 1.22 Å |
| Electron Affinity | Unknown |
| Electronegativity | Unknown |
| Ionization Energy | Unknown |
What are the Uses of Copernicium
Copernicium is not a natural element, but can only be synthesized in the laboratory, making it useful for nothing more than scientific research [1]. The instability of the element makes it unsuitable for any use in everyday life [2, 4].
Interesting Facts
- The symbol Cp was first recommended for the metal but was rejected as it had already been used for lutenium till 1949. So for more than a decade, it was known as element 112 [1, 2].
- Roentgenium, Bohrium, Darmstadtium, Hassium, and Meitnerium were also discovered at the same time as Cn [2].
- Cn is the heaviest element in existence, specifically 200 times heavier than hydrogen [2].
Copernicium Cost
As Cn has a short half-life, it is not available outside of nuclear laboratory production [7].
- References
- https://www.rsc.org/periodic-table/element/112/copernicium
- http://www.elementsdatabase.com/Copernicium-Cn-112-element/
- https://www.americanelements.com/copernicium.html
- http://byjus.com/chemistry/copernicium/
- https://environmentalchemistry.com/yogi/periodic/Cn.html#Physical
- http://metals.comparenature.com/en/atomic-structure-of-copernicium/model-71-5
- https://en.wikipedia.org/wiki/Prices_of_elements_and_their_compounds#cite_note-1
- https://www.raci.org.au/document/item/416