Lanthanum
What is Lanthanum
Lanthanum (pronunciation: LAN-the-nem) [3] is a soft, silvery rare-earth metal belonging to the lanthanoids family and represented by the chemical symbol La [1, 2]. Its main isotopes include the most abundant 139La (with 99.91% natural abundance) and the most stable 138La, the radioactive isotope with a half-life period of 1.06 X 1026 years [1, 4].
Where can Lanthanum be Found
The element naturally occurs in bastnaesite and monazite, making up 38% and 25% of the rare-earth minerals respectively [1]. Isolation of lanthanum from these minerals involves solvent extraction and ion-exchange separation techniques [1]. The metal can be produced through calcium reduction of anhydrous fluoride [1]. The top 3 lanthanum producing countries include China, Russia, and Malaysia while the top 3 countries with the largest lanthanum reserves are China, CIS Countries, and the USA [1].
History
Origin of its Name: The name of the element is derived from the Greek word ‘lanthanein’, which means to lie hidden, as its existence in cerium oxide was long undetected [1].
Who discovered it: The Swedish chemist Carl Gustaf Mosander is credited with the discovery of lanthanum [1].
When and How was it Discovered
In January 1839, lanthanum was extracted by Carl Mosander while working on his sample of cerium at the Karolinska Institute in Sweden [1]. He took powdered cerium nitrate and treated it with dilute nitric acid [5]. He observed that some of the nitrate powder dissolved in the acid, and then he decided to isolate the mixture from the precipitate by using heat and sodium oxalate [5]. The pale brick-colored oxide obtained was given the name lanthana [3, 5].
A student at the Karolinska Institute, Axel Erdmann discovered the element independently from a new mineral found in a Norwegian fjord [1, 5]. The new mineral was named mosandrite after Carl Mosander who produced impure lanthanum from anhydrous cerium chloride [5]. In 1923, pure lanthanum was obtained by Kremers and Stevens through electrolysis of fused halides [5].
Lanthanum Identification |
|||
| Atomic number | 57 [1] | ||
| CAS number | 7439-91-0 [1] | ||
| Position in the periodic table | Group | Period | Block |
| Lanthanides [1] | 6 [1] | d [1] | |
Properties and Characteristics of Lanthanum
General Properties |
||||||||||||||||
| Atomic mass | 138.905 amu [1] | |||||||||||||||
| Relative atomic mass | 138.905 [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery-white [1, 5] | |||||||||||||||
| Melting point/freezing point | 920 °C, 1688 °F [1] | |||||||||||||||
| Boiling point | 3464 °C, 6267 °F [1] | |||||||||||||||
| Density | 6.15 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 5] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 363 MPa [6] | |||||||||||||||
| – Mohs | 2.5 [6] | |||||||||||||||
| – Vickers | 491 MPa [6] | |||||||||||||||
| Electrical conductivity | 1.6X106 S/m [6] | |||||||||||||||
| Thermal (heat) conductivity | 13 W/(m K) [6] | |||||||||||||||
| Specific heat | 195 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 27.9 GPa [1] | |||||||||||||||
| Shear modulus | 14.3 GPa [1] | |||||||||||||||
| Young’s modulus | 36.6 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | 5.09X 10-8 | 2.02X 10-5 | 1.81X 10-3 | 5.96X 10-2 | 0.976 | 9.61 | 64.7 | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | +2, +3 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance | Half-life | Mode of decay | |||||||||||
| 138La | 137.907 | 0.09 | 1.06 X 1011 y | – | ||||||||||||
| 139La | 138.906 | 99.91 | – | – | ||||||||||||
Atomic Data of Lanthanum (Element 57)
| Valence electrons | 3 [6] | |||||||
| Quantum numbers | ||||||||
| – n | 4 [7] | |||||||
| – ℓ | 3 [7] | |||||||
| – mℓ | -3 [7] | |||||||
| – ms | +1/2 [7] | |||||||
| Electron configuration (noble gas configuration) | [Xe] 5d16s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 57 [5] | |||||||
| – Number of neutrons | 82 [5] | |||||||
| – Number of protons | 57 [5] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.43 Å [1] | |||||||
| – Covalent radius | 1.94 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.10 [1] | |||||||
| Electron affinity | 45.35 kJ mol-1 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 538.089 | 1067.031 | 1850.328 | 4819.44 | 5943.5 | – | – | – | |
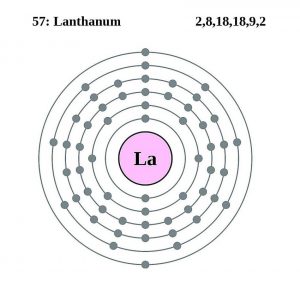
Lanthanum Electron Configuration (Bohr Model)
What is Lanthanum Used for
Although lanthanum metal is not commercially used, its alloys may have some uses [1]:
- An alloy of lanthanum and nickel is used for hydrogen storage in hydrogen-fueled vehicles [1]. It is used in large amounts in the anode (positively charged electrode) of NiMH (nickel metal hydride) batteries that are found in hybrid cars [1, 5].
- Lanthanum is a constituent element in misch metal (an alloy of rare earth metals) that is used in flints for producing igniting sparks in cigarette lighters [1].
- Compounds with lanthanum are extensively used in cinema projection, studio lighting, and other carbon lighting applications [1].
- Lanthanum (III) oxide (La2O3) is used in producing high-quality optical glasses, telescope and camera lenses because it has low dispersion, high refractive index, and it imparts high alkali resistance [1, 5], producing a clear, sharp image.
- It is used as a catalyst in petroleum industries for cracking the long chain hydrocarbons [5].
- Ionic lanthanum (La3+) is used as a tracer and marker of Ca2+ spikes in blood, and studies have shown that radioactive lanthanum and its compounds may be used in treating cancer [1].
- Lanthanum carbonate (fosrenol) is useful for reducing phosphate levels in people affected by kidney disease [5].
Possible Health Effects
Lanthanum and its compounds, although not known to play any biological role in humans, can be a little or moderately toxic and should be handled with caution and proper protection [1].
Interesting Facts
- The element rapidly oxidizes in air and is easily burned when ignited [1].
- It is sometimes visually represented with an image of a camera lens in reference to its use in cameras [1].
How much does Lanthanum Cost
The price of 100 grams of pure lanthanum is around $800 while its price in bulk ranges between $6,000 and $6,500 [5, 8].
- References
- http://www.rsc.org/periodic-table/element/57/lanthanum
- https://pubchem.ncbi.nlm.nih.gov/compound/lanthanum#section=Top
- https://education.jlab.org/itselemental/ele057.html
- https://www.webelements.com/lanthanum/isotopes.html
- https://www.chemicool.com/elements/lanthanum.html
- http://periodictable.com/Elements/057/data.html
- http://chemistry-reference.com/q_elements.asp?language=en&Symbol=La
- http://hobart.k12.in.us/ksms/PeriodicTable/lanthanum.htm







