Nihonium
What is Nihonium
Nihonium (pronunciation nee-hone-ee-em [2]), represented by the chemical symbol Nh, is a synthetic, radioactive, transuranium metal [1, 10, 11]. Of its two known isotopes, Nh 284 and Nh 286 [1], the latter is more stable with a half-life of 20 seconds [2].
Where Is It Found
The super heavy element cannot be found naturally on earth. A tiny amount has been synthesized in labs [1, 3].
History
Origin of Its Name: It is named after the Japanese name for Japan [1].
Who Discovered It: Scientists from The Institute of Physical and Chemical Research (RIKEN) in Japan [1].
How Was Nihonium Discovered
The first two atoms of the element were produced on July 23, 2004, at RIKEN by accelerating zinc ions to make them reach 10% of the speed of light and then allowing them to strike a bismuth target. The atoms were of nihonium 278 that quickly underwent alpha decay to dubnium 262 [2, 3].
Nihonium Identification |
|||
| Atomic Number | 113 [1] | ||
| CAS Number | 54084-70-7 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 13 [1] | 7 [1] | p [1] | |
Properties and Characteristics of Nihonium
General Properties |
|
| Atomic mass | 286 atomic mass units [1] |
| Atomic weight | 286 [1] |
Physical Properties |
|
| Color/appearance | Unknown [4,5] |
| Melting point/freezing point | Unknown [1] |
| Boiling point | Unknown [1] |
| Density | Unknown [1] |
| State of matter at room temperature (normal phase) | Solid (estimated) [1,3] |
Chemical Properties |
|
| Oxidation state/Oxidation number | [+1], [+2], [+3], [+5] [1] |
| Reactivity | Unknown [8] |
Atomic Data of Nihonium (Element 113)
| Valence electrons | 7s27p1 [9] (as predicted depending on its position in periodic table) | ||||||
| Electron configuration (noble gas configuration) | [Rn] 5f146d107s27p1 [1] | ||||||
| Atomic structure | |||||||
| – Number of Electrons | 113 [3] | ||||||
| – Number of Neutrons | 113 [3] | ||||||
| – Number of Protons | 173 [3] | ||||||
| Energy levels [3] | |||||||
| – First Energy Level | 2 | ||||||
| – Second Energy Level | 8 | ||||||
| – Third Energy Level | 18 | ||||||
| – Fourth Energy Level | 32 | ||||||
| – Fifth Energy Level | 32 | ||||||
| – Sixth Energy Level | 18 | ||||||
| – Seventh Energy Level | 3 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | Unknown [1] | ||||||
| – Covalent Radius | 1.36 Å [1] | ||||||
| Electronegativity | Unknown [1] | ||||||
| Ionization energy
(kJmol-1) [1] |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| – | – | – | – | – | – | – | |
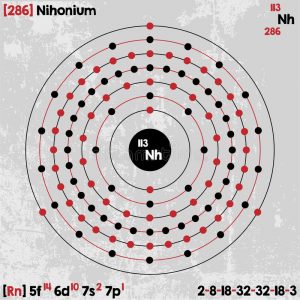
Nihonium Electron Configuration (Bohr Model)
Nihonium Uses
The minuscule amount of production of the metal restricts its use to research purposes only [1].
Is It Dangerous
The highly radioactive nature makes it potentially harmful [1, 3].
Interesting Facts
- It was temporarily given the name ununtrium (symbol Uut [4]) representing its atomic number before being officially named by IUPAC on 28th November 2016 along with moscovium, tenessine and oganesson [6, 7].
- The name Japonium was considered for the element by the researchers to emphasize the Japan connection, but the proposal was dismissed since the word Jap is insulting to the Japanese [12].
Nihonium (Ununtrium) Cost
Unknown as it is not available commercially [3].
- References>
- http://www.rsc.org/periodic-table/element/113/nihonium
- https://education.jlab.org/itselemental/ele113.html
- https://www.chemicool.com/elements/nihonium.html
- https://www.webelements.com/nihonium/
- http://periodictable.com/Properties/A/Color.html
- https://iupac.org/iupac-announces-the-names-of-the-elements-113-115-117-and-118/
- https://www.npr.org/sections/thetwo-way/2016/06/09/481410462/hello-nihonium-scientists-name-4-new-elements-on-the-periodic-table
- https://www.webelements.com/nihonium/chemistry.html
- https://www.schoolmykids.com/learn/interactive-periodic-table/nh-nihonium/
- https://www.livescience.com/41416-facts-about-ununtrium.html
- https://www.britannica.com/science/element-113
- https://mainichi.jp/english/articles/20160609/p2a/00m/0na/010000c






it’s very much helpful for students