Phosphorus
What is Phosphorus
Phosphorus (pronunciation: FOS-fer-es) is a highly-reactive element classified as a non-metal represented by the chemical symbol P [1, 2, 3]. It mainly exists in its two allotropic forms, including the waxy, white solid and the non-crystalline, solid red that is obtained by heating white phosphorus [3, 4, 5].
Isotopes
There are 24 isotopes of phosphorus, ranging from 24P to 47P out of which 31P is stable [6]. While most of the radioactive isotopes are short-lived with half-life periods less than 2.5 minutes, 33P and 32P are the longest-lived with half-lives of 25.34 days and 14.263 days respectively [6]. The least stable among the 24 isotopes of 15P is 25P characterized by a half-life period less than 30 nanoseconds [6].
Where is Phosphorus Found
Phosphorus does not occur freely in nature, but it forms compounds with other elements in minerals [1]. It is mainly obtained from phosphate rock, including the apatite phosphate mineral [1].
White phosphorus is commercially produced by treating phosphate rock with silica and carbon in a fuel-fired or electric furnace [1]. Phosphorus is produced as a vapor, which is collected under water [1]. Red phosphorus is manufactured by heating the white form to 250 °C in an inert atmosphere [1].
The top 3 phosphorus producing countries are China, Mexico, and Morocco while the top 3 phosphorus reserve holders include Morocco, China, and the USA [1].
History
Origin of its Name: It is derived from ‘phosphoros’, Greek for the bringer of light [1]
Who discovered it: The element was discovered by the German merchant and alchemist Hennig Brand [1, 2].
When and How was it Discovered
In 1669, Hennig Brand discovered phosphorus at Hamburg by boiling urine and then heating the residue, which produced phosphorus vapor [1]. Then, he distilled the phosphorus vapor and collected it after condensing it in the water [1]. Hennig Brand decided to keep his discovery secret because he thought that he had produced the Philosopher’s Stone which could be used for turning metals into gold [1].
Since Hennig needed money, he sold it to Daniel Kraft who displayed it around Europe, capturing the attention of Robert Boyle [1]. He studied phosphorus systematically and found out its characteristics [1].
Phosphorus Identification |
|||
| Atomic number | 15 [1] | ||
| CAS number | 7723-14-0 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 15 [1] | 3 [1] | p [1] | |
Properties and Characteristics of Phosphorus
General Properties |
||||||||||||||||
| Relative atomic mass | 30.974 [1] | |||||||||||||||
| Atomic mass | 30.974 amu [1] | |||||||||||||||
| Molar mass | 30.9737620 ± 0.0000020 g/mol [7] | |||||||||||||||
| Molecular weight | 30.974 g/mol [3] | |||||||||||||||
| Allotropes | White (yellow), Red, Black [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Usually, white or pale yellow [5] | |||||||||||||||
| Melting point/freezing point | 44.15 °C, 111.47 °F [1] | |||||||||||||||
| Boiling point | 280.5 °C, 536.9 °F [1] | |||||||||||||||
| Density | 1.823 g cm-3 (white) [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 5] | |||||||||||||||
| Electrical Conductivity | 1 X 107 S/m [8] | |||||||||||||||
| Charge | -3 [9] | |||||||||||||||
| Thermal (heat) conductivity | 0.236 W/(m K) [8] | |||||||||||||||
| Specific heat | 769 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 10.9 GPa (red), 4.9 GPa (white) [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | – | – | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | -3, -2, -1, +1, +2, +3, +4, +5 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 31P | 30.974 | 100 | – | – | ||||||||||||
Atomic Data of Phosphorus (Element 15)
| Valence electrons | 5 [10] | |||||||
| Quantum numbers | ||||||||
| – n | 3 [10] | |||||||
| – ℓ | 1 [10] | |||||||
| – mℓ | 1 [10] | |||||||
| – ms | +1/2 [10] | |||||||
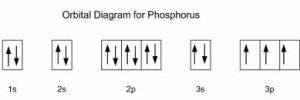
| Electron configuration (noble gas configuration) | [Ne] 3s23p3 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 15 [11] | |||||||
| – Number of neutrons | 16 [11] | |||||||
| – Number of protons | 15 [11] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 1.80 Å [1] | |||||||
| – Covalent radius | 1.09 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 2.19 [1] | |||||||
| Electron affinity | 72.037 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 1011.812 | 1907.467 | 2914.118 | 4963.582 | 6273.969 | 21267.395 | 25430.64 | 29871.9 | |

Phosphorus Electron Configuration (Bohr Model)
What is Phosphorus Used for
- Its allotropic white form is used in incendiary bombs and flares [1].
- Red phosphorus is used in manufacturing safety matches and is stuck on the matchboxes so that the matches can be lit by striking against them [1, 5].
- Phosphorus compounds are generally used in fertilizers [1, 5].
- Phosphate ores are an important source of ammonium phosphate [1]. The ores are converted into phosphoric acids and then into ammonium phosphate [1].
- It is also an essential ingredient in the manufacture of steel, phosphor bronze, and some detergents [1, 5]. However, its use in detergents has now been phased out in several countries because it can accumulate in natural water supplies and induce the growth of unwanted algae [1].
- It is used in manufacturing fine chinaware and special glasses [1].
- Phosphorus is one of the materials used for producing LEDs (light-emitting diodes) [5].
Toxicity and Health Effects
An excess of phosphate in the human body can cause soft tissue, hardening of organs, or diarrhea [12]. It can also affect your body’s mechanism of utilizing other essential minerals like zinc, calcium, magnesium, and iron [12].
While too much phosphorus is toxic for your health, too little of it can weaken your teeth and bones, as well as cause anxiety, fatigue, and loss of appetite [12].
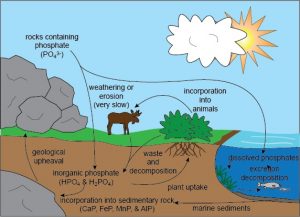
It is an essential nutrient for plants, which help in moving it through the biosphere, lithosphere, and hydrosphere and this cycle is known as the phosphorus cycle [5].
Interesting Facts
- The average amount of phosphates in the human body, mostly in the teeth and bones, is estimated at 750 g (26.5 ounces) [4].
- A 2013 study has shown that the phosphorus in the Earth’s crust may have been brought by meteorites [4].
- High phosphorus levels in the blood is a warning sign for heart diseases [4].
- Since white phosphorus exists as molecules consisting of four atoms arranged in a tetrahedral structure, its graphical representation indicates this tetrahedral shape with a ball-and-stick model [1].
Cost of Phosphorus
The price of pure phosphorus is $0.3 per gram [5].
- References
- http://www.rsc.org/periodic-table/element/15/phosphorus
- https://education.jlab.org/itselemental/ele015.html
- https://pubchem.ncbi.nlm.nih.gov/compound/5462309#section=Top
- https://www.livescience.com/28932-phosphorus.html
- https://www.chemicool.com/elements/phosphorus.html
- https://education.jlab.org/itselemental/iso015.html
- https://www.webqc.org/molecular-weight-of-Phosphorus.html
- http://periodictable.com/Elements/015/data.html
- Lenntech.com
- http://chemistry-reference.com/q_elements.asp?Symbol=P
- https://hobart.k12.in.us/ksms/PeriodicTable/phosphorus.htm
- https://www.healthline.com/health/phosphorus-in-diet#toomuch-phosphorus