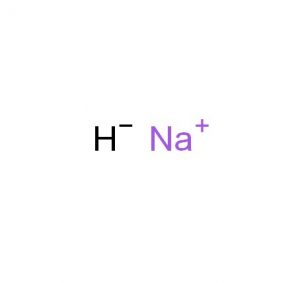
Sodium Hydride
Sodium hydride represented by the chemical formula NaH or HNa [1] is a highly reactive inorganic ionic compound and a strong base. Due to its high reactivity, it is usually available as a gray solid dispersed in mineral oil that allows safe handling [2].
Sodium Hydride Identification |
|
| CAS Number | 7646-69-7 [1] |
| PubChem CID | 24758 [1] |
| ChemSpider ID | 23144 [3] |
| EC Number | 231-587-3 [1] |
How to Make It
Sodium hydride can be prepared by a direct reaction between hydrogen gas and sodium metal [2].
2 Na + H2 = 2NaH
Properties and Characteristics of Sodium Hydride
General Properties |
||
| Molar mass/molecular weight | 23.998 g/mol [1] | |
Physical Properties |
||
| Color/appearance | Silvery to whitish powder [1] | |
| Melting point/freezing point | 800°C, 1472°F [2] | |
| Boiling point | N/A [4] | |
| Flash point | N/A [1] | |
| Density | 1.396 g cm-3 [4] | |
| State of matter at room temperature (normal phase) | Solid [1] | |
Chemical Properties |
||
| Solubility in water | Reacts [5] | |
| pH | >7 (basic) [2] | |
Atomic Properties |
||
| Crystal structure | Octahedral [2] | |
Prominent Reactions
NaH reacts violently with water to produce sodium hydroxide and hydrogen gas that gets ignited by the heat of the reaction [5].
NaH + H2O = NaOH + H2
Alcohols react with sodium hydride to produce alkoxide ions. In the below reaction, ethanol reacts with NaH forming sodium ethoxide and hydrogen [10].
CH3CH2OH + NaH = CH3CH2O–Na+ + H2
Similarly, methanol reacts with sodium hydride producing the sodium salt of methanol and hydrogen gas [8].
CH3OH + NaH = CH3ONa + H2
Sodium Hydride Uses
- As a powerful reducing and deprotonating agent for many organic reactions [2].
- As a desiccant or drying agent for lab chemicals [2].
- Its application as a hydrogen storage agent in fuel cell vehicles is being explored due to its ability to release hydrogen [2].
- As a strong base, it finds application in organic and inorganic fine chemical preparation [6].
Is It Dangerous
NaH is a severe irritant to eyes, skin and respiratory system causing burns and is harmful if ingested. It is a highly inflammable and corrosive compound. Precaution must be taken against an explosion hazard if it comes in contact with water [2, 7]. When it is heated to the point of decomposition, it emits highly poisonous fumes of oxides of sodium [1]. After a reaction, unwanted sodium hydride should be destroyed by careful quenching or disposal [9].
Interesting Facts
- In spite of its high basicity, NaH is not nucleophilic [12].
Sodium Hydride (NaH) Price
It costs around $30-50 per kilogram. The price may vary with suppliers [11].
- References
- Sodium Hydride (NaH) – Pubchem.ncbi.nlm.nih.gov
- Sodium hydride Formula – Sodium hydride Uses, Properties, Structure and Formula – Softschools.com
- Sodium Hydride – Chemspider.com
- Sodium Hydride – Americanelements.com
- Sodium Hydride – Onlinelibrary.wiley.com
- Sodium Hydride – Chemicalland21.com
- Sodium Hydride – Cameochemicals.noaa.gov
- What is the reaction between sodium hydride and methanol? – Answers.yahoo.com
- Sodium Hydride – Sites.chemengr.ucsb.edu
- Alcohols, Phenols, Ethers, Epoxides, Thiols, &Sulfides – Chem.latech.edu
- Sodium hydride price – Alibaba.com
- Illustrated Glossary of Organic Chemistry – Chem.ucla.edu