Titanium
What is Titanium
Titanium (pronunciation: ti-TAY-nee-em) is a hard, shiny, strong, and lightweight element classified as a transition metal and represented by the chemical symbol Ti [1, 2, 3]. As compared to the other ferromagnetic elements, titanium is a weakly magnetic metal that shows its magnetic characteristics in the presence of external magnetic fields [4].
Isotopes
There are five stable naturally-occurring isotopes of titanium, including Titanium-46, Titanium-47, Titanium-48, Titanium-49, and Titanium-50, with Titanium-48 having the greatest natural abundance of 73.8% [5]. Titanium is characterized by 21 radioisotopes out of which the most stable are Titanium-44, Titanium-45, Titanium-51, and Titanium-52 with half-lives of 60 years, 184.8 minutes, 5.76 minutes, and 1.7 minutes respectively [5].
Where is Titanium Found
As the 9th most abundant element in the Earth’s crust, titanium is commonly found in igneous rocks and their sediments [1]. It is present in minerals like rutile (titanium dioxide), ilmenite (titanium-iron oxide), and sphene (calcium titanium nesosilicate), as well as in titanate (titanium oxide) and several iron ores [1].
It is commercially extracted from its ore through reduction of titanium (IV) chloride using either sodium or magnesium [1]. Titanium (IV) oxide is obtained from ilmenite by using either chlorine (chloride process) or sulfuric acid (sulfate process) [1].
The top 3 titanium producers are Canada, Australia, and South Africa while the top 3 titanium reserve holding countries include China, Australia, and India [1].
History
Origin of its Name: The name is derived from the word ‘Titan’, which in Greek mythology means “the sons of the Goddess Earth” [1, 2].
Who discovered it: Titanium was found by the British mineralogist and clergyman William Gregor [1].
When and How was it Discovered
In 1791, the titanium mineral menaccanite was found in Cornwall by William Gregor [1]. He studied it and inferred that it consisted of iron oxide along with a new metal, which he reported to the Royal Geological Society [1].
The German chemist Martin Heinrich Klaproth, in 1795, analyzed a red mineral ore called Schörl [1]. It was the oxide of an unknown element which Klaproth named titanium [1]. After learning about Gregor’s discovery, Klaproth examined menaccanite and confirmed that it was made up of titanium [1].
M.A. Hunter, the metallurgist from New Zealand, produced pure titanium metal from titanium tetrachloride and sodium metal in 1910 [1].
Titanium Identification |
|||
| Atomic number | 22 [1] | ||
| CAS number | 7440-32-6 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 4 [1] | 4 [1] | d [1] | |
Properties and Characteristics of Titanium
General Properties |
||||||||||||||||
| Relative atomic mass | 47.867 [1] | |||||||||||||||
| Atomic mass | 47.867 amu [1] | |||||||||||||||
| Molar mass | 47.8670 g/mol [6] | |||||||||||||||
| Molecular weight | 47.867 g/mol [7] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery-white [8] | |||||||||||||||
| Melting point/freezing point | 1670 °C, 3038 °F [1] | |||||||||||||||
| Boiling point | 3287 °C, 5949 °F [1] | |||||||||||||||
| Density | 4.506 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 8] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 715 MPa [9] | |||||||||||||||
| – Mohs | 6 [9] | |||||||||||||||
| – Vickers | 970 MPa [9] | |||||||||||||||
| Electrical Conductivity | 2.5 X 106 S/m [9] | |||||||||||||||
| Charge | +3, +4 [10] | |||||||||||||||
| Thermal (heat) conductivity | 22 W/(m K) [9] | |||||||||||||||
| Specific heat | 524 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | Unknown [1] | |||||||||||||||
| Shear modulus | 43.8 GPa [1] | |||||||||||||||
| Young’s modulus | 115.7 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | 9.69 X 10-9 | 7.44 X 10-6 | 1.06 X 10-3 | 0.0493 | 0.978 | 10.6 | 76.9 | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | -1, (+1), +2, +3, +4 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 46Ti | 45.953 | 8.25 | – | – | ||||||||||||
| 47Ti | 46.952 | 7.44 | – | – | ||||||||||||
| 48Ti | 47.948 | 73.72 | – | – | ||||||||||||
| 49Ti | 48.948 | 5.41 | – | – | ||||||||||||
| 50Ti | 49.945 | 5.18 | – | – | ||||||||||||
Atomic Data of Titanium (Element 22)
| Valence electrons | 4 [11] | |||||||
| Quantum numbers | ||||||||
| – n | 3 [12] | |||||||
| – ℓ | 2 [12] | |||||||
| – mℓ | -1 [12] | |||||||
| – ms | +1/2 [12] | |||||||
| Electron configuration (noble gas configuration) | [Ar] 3d24s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 22 [8] | |||||||
| – Number of neutrons | 26 [8] | |||||||
| – Number of protons | 22 [8] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.11 Å [1] | |||||||
| – Covalent radius | 1.48 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.54 [1] | |||||||
| Electron affinity | 7.622 kJ mol-1 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 658.813 | 1309.837 | 2652.546 | 4174.651 | 9581 | 11532.89 | 13585.1 | 16441.1 | |
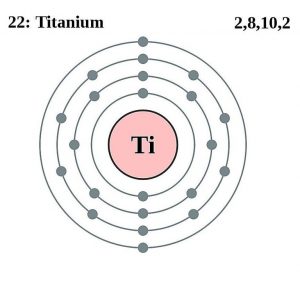
Titanium Electron Configuration (Bohr Model)
What is Titanium Used for
- Titanium forms important alloys with other metals, including molybdenum, iron, and aluminum, that are used in missiles, aircraft, and spacecraft due to their low density and high-temperature resistance [1, 8].
- Although it is as strong as steel, it has a much lesser density because of which it is commonly used in laptops, golf clubs, crutches, and bicycles [1, 8].
- Since they resist corrosion, titanium pipes are used in power plant condensers [1].
- As it has excellent corrosion resistance in seawater, it is widely used for propeller shafts, hulls of ships, submarines, and in desalination plants [1, 8].
- Titanium has many surgical applications, including its use in hip joint replacements and tooth implants [1, 8].
- Since its pigment has great covering power, it is used in plastics, artists’ paint, house paint, enamels, and paper [1].
- It is used in solar observatories because of its excellent infrared-radiation reflective properties [1].
- Titanium oxide is an essential ingredient in sunscreens, used for protecting the skin from UV light [1].
Toxicity and Health Effects
While pure titanium is considered non-toxic, exposure to large amounts of titanium dioxide dust may be hazardous [1]. Although the safety of nano-TiO2 is unclear, studies have shown that it may damage the liver, kidney, brain, and lung functions when it is inhaled or taken in high doses [13]. It can also be toxic to embryos in development [13]. Titanium is not known to play any biological role [1].
Interesting Facts
- Every igneous rock (that is formed due to the cooling and solidification of lava) consists of titanium [3].
- According to Boeing, the 737 Dreamliner comprises 15 percent titanium [3].
- A 2011-satellite-image revealed titanium-rich rocks on the moon’s surface [3].
- The graphical representation of titanium indicates an early votive statue based on the Titans in Greek mythology [1].
Titanium Price
The cost of pure titanium is about $6.61 per gram [8].
- References
- http://www.rsc.org/periodic-table/element/22/titanium
- https://education.jlab.org/itselemental/ele022.html
- https://www.livescience.com/29103-titanium.html
- https://terpconnect.umd.edu/~wbreslyn/magnets/is-titanium-magnetic.html
- https://education.jlab.org/itselemental/iso022.html
- https://www.webqc.org/molecular-weight-of-Ti.html
- https://pubchem.ncbi.nlm.nih.gov/compound/titanium
- https://www.chemicool.com/elements/titanium.html
- http://periodictable.com/Elements/022/data.html
- http://www.ochemonline.com/Ions
- http://butane.chem.uiuc.edu/pshapley/GenChem2/Intro/2.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Ti
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5540742/