Acryloyl Chloride
- Acryloyl Chloride Definition
- Acryloyl Chloride Identification Facts
- Acryloyl Chloride Molecular Formula
- Acryloyl Chloride Synonyms
- Acryloyl Chloride Chemical Structure
- Acryloyl Chloride Preparation in Laboratory
- Acryloyl Chloride Properties
- Thermodynamic Properties
- Acryloyl Chloride Uses
- Acryloyl Chloride MSDS
- Acryloyl Chloride Organic Synthesis
- Acryloyl Chloride Reactions
Acryloyl Chloride Definition
It is a light-yellow, combustible liquid (at STP) with a pungent odor.
Acryloyl Chloride Identification Facts
The identification facts of this chemical are as follows:
CAS No.
The CAS No. of this compound is 814-68-6 .
PubChem
The PubChem number for this chemical is 399362.
ChemSpider
The ChemSpider no. for this compound is 12588.
EC No.
The EC No for this substance is 212-399-0.
Acryloyl Chloride Molecular Formula
The molecular formula for this liquid is C3H3ClO.
Picture 1 – Acryloyl Chloride
Source – 0.tqn.com
Acryloyl Chloride Synonyms
This chemical is also known by other names, such as:
- 2-propenoyl Chloride
- Acrylic Acid Chloride
- Propenoyl Chloride
- Prop-2-enoyl Chloride
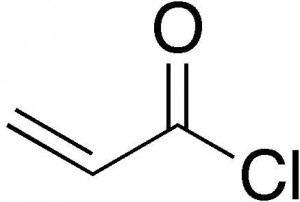
Acryloyl Chloride Chemical Structure
Take a look at the chemical structure of this substance:
Acryloyl Chloride Preparation in Laboratory
This chemical can be prepared in the laboratory by reacting Acrylic acid with Benzoyl Chloride or with Thionyl Chloride. If a small quantity of an inhibitor like Hydroquinone is added during the preparation, light-induced polymerization of Acryloyl Chloride can be avoided.
Acryloyl Chloride Properties
Some of the main physical and chemical properties of this compound are as follows:
Molar Mass
Its molecular weight (mw) is 90.51 g mol−1 (or 90.509 g/mol).
Density
The density of this substance is 1.114 g/cm³.
Boiling Point
The boiling point (bp) of this chemical is 74°C.
Melting Point
Its melting point (mp) is -72°C.
Vapor Density
Its vapor density is greater than 1.
Vapor Pressure
The vapor pressure of this liquid is 99.78 mmHg (STP). It is also represented as 1.93 psi at 20 °C.
Specific Gravity
It has a specific gravity of 1.114.
Refractive Index
This compound has a refractive index of 1.4350.
Stability
It is stable in nature but reacts violently with water. It is not compatible with alcohols, strong bases and strong oxidizing agents. It is sensitive to light and is highly inflammable.
Thermodynamic Properties
Go through the thermodynamic properties of this substance.
Critical Temperature
Its critical temperature is 546 K.
Critical Pressure
Its critical pressure is 4.83 MPa (STP).
Flash Point
The flash point of this liquid is 16.11°C.
Solubility
It is soluble in water.
Stabilizer
Commercial liquid often uses <210 ppm MEHQ (Monomethyl Ether of Hydroquinone) as stabilizer.
Reactivity
This chemical reacts violently with water producing Acrylic acid. It reacts with Sodium salts of Carboxylic acids forming Anhydrides. These are consistent with common reactions of Chloride acids. It reacts with alcohols forming esters. It produces amides on reacting with amines.
The chemical is incompatible with strong bases, oxidizing agents and alcohols.
Acryloyl Chloride Uses
This chemical has a number of uses. Some of its main uses can be summarized as follows:
- It is also used widely in the manufacture of Acrylate monomers and polymers.
- It is an excellent substrate for cross-metathesis.
- Because of its high reactivity, this chemical is used for manufacturing new polymers by polymer-analogous transformations when polymerization of the associated monomers is not easy.
Acryloyl Chloride MSDS
Go through the Material Safety Data Sheet (MSDS) of this compound:
Inflammability
It is an easily inflammable liquid. This compound can result in burns when inhaled, ingested or accidentally put in contact with skin.
Toxicity
It is a highly toxic liquid. It may be fatal if swallowed, inhaled or absorbed through skin. It causes burns and is irritating to eyes and respiratory system. It is also damaging to mucous membranes.
Transportation
As it is a highly flammable liquid, it belongs to the highest risk category (great danger) during transportation. It belongs to Hazard Class 3.0 and Packing Group I. The material should be transferred in bond and ground containers. Explosion proof equipment and spark-proof tools should be used during transfer.
In case of accidental spillage, the spillage of the inert material should immediately be absorbed and put in a suitable container – as per precautions in the Protective Equipment section. All sources of ignition should be removed. If vapors are produced, a vapor suppressing foam may be used for reducing them.
The substance should be handled properly, as it is a Lachrymator (tear-producing gas).
Personal Safety
It is important to keep this liquid away from sources of ignition. The compound should be used carefully as it is also a lachrymator, which means it causes eyes to emit tears.
People using this material should wear appropriate protective gear. There should be provisions for proper eye and face protection. The safety gear commonly used for protection involve
- Safety glasses (for eyes)
- Gloves (for the hands)
- Lab coat (for overall protection of the body)
- Respiratory equipments (for good ventilation)
First Aid
In case of eye or skin exposure, plenty of water should be used to wash impurities off the affected area for fifteen minutes. Medical advice should urgently be sought. Contaminated clothes should be immediately taken off and washed thoroughly before wearing them again.
If a person suffers from accidental ingestion of this substance and is found to be conscious, he or she should be provided with 2-4 cups filled with water or milk. Medical assistance should immediately be sought.
In case of accidental inhalation of this substance, an affected person should immediately be removed to a fresh, airy place. He or she should seek medical advice as soon as possible. If the person is not found to be breathing, artificial respiration should be used. Oxygen should be provided in case of respiratory difficulties.
Storage Instructions
The chemical should be stored in a tightly closed container in a cool, dry, well-ventilated area. It should ideally be kept refrigerated at temperatures under 4°C/39°F. It should be kept away from sparks, flame, heat and other sources of ignition. The container should be kept closed when the chemical is not in use.
Acryloyl Chloride Organic Synthesis
One of the main applications of this chemical is in organic synthesis. The compound is used in organic synthesis to introduce Acrylic moieties (one of two equal parts) into other compounds.
Acryloyl Chloride Reactions
This compound produces a violent reaction with water-producing Acrylic Acid. It forms anhydrides on reacting with sodium salts of carboxylic acids. It reacts with alcohols and amines to form esters and amides respectively.
- References
- http://pubs.acs.org/doi/abs/10.1021/ol9021386
- https://www.wolframalpha.com/input/?i=acryloyl+chloride
- http://chemistry.about.com/od/factsstructures/ig/Chemical-Structures—A/Acryloyl-Chloride.htm
- http://www.chemicalbook.com/ChemicalProductProperty_EN_CB4854427.htm





What is the heat of reaction of acryloylchloride with alcohols ?