Chromium (III) Oxide
Chromium (iii) oxide, commonly called chromia and referred to as oxo(oxochromiooxy)chromium in IUPAC nomenclature, is an inorganic compound containing chromium and oxygen represented by the chemical formula Cr2O3 [1]. It naturally occurs as the mineral eskolaite and is a highly insoluble compound with high thermal stability, having the properties of both an acid and a base [2, 3, 4].
How is Chromium (III) Oxide Prepared
It is produced through the reduction of sodium dichromate using sulfur at high temperatures indicated by the redox reaction [6]:
Na2Cr2O7 + S → Na2SO4 + Cr2O3
Ammonium dichromate, on heating, decomposes into chromium (iii) oxide, water, and nitrogen [7]:
(NH4)2Cr2O7 → Cr2O3 + N2 + 4H2O
Chromium (iii) carbonate is heated to form chromium (iii) oxide and carbon dioxide:
Cr2(CO3)3 → Cr2O3 + 3CO2
Reaction with Other Compounds
Chromium (III) Oxide with Hydrochloric Acid
It reacts with dilute hydrochloric acid to form chromium (iii) chloride and water [8]:
Cr2O3 + 6HCl → 2CrCl3 + 3H2O
Chromium (III) Oxide with Hydrogen Sulfide
When chromium (iii) oxide reacts with gaseous hydrogen sulfide, it yields chromium (iii) sulfide and water:
Cr2O3 + 3H2S → Cr2S3 + 3H2O
Properties and Characteristics of Chromium (III) Oxide
General Properties |
|
| Molar Mass/Molecular Weight | 151.989 g/mol [1, 3] |
Physical Properties |
|
| Color and Appearance | Dry, light or dark green, fine crystalline powder [1, 3] |
| Melting Point | 2,435 °C, 4,415 °F [1] |
| Boiling Point | 4,000 °C, 7,230 °F [1] |
| Density | 5.22 g cm-3 [1, 3] |
| State of matter at room temperature | Solid [1, 3] |
| Solubility | Insoluble in acetone and alcohol, very slightly soluble in alkalies and acids [1] |
| Solubility in Water | Practically insoluble, 2.96 µg/L (20 °C, pH 8), 3.13 µg/L (20 °C, pH 6) [1] |
| Magnetic Susceptibility (χ) | 1960.0 X 10-6 cm3/mol |
Atomic Properties |
|
| Crystal Structure | Hexagonal |
Uses
- Because of its stability, chromium (iii) oxide is used as the green pigment in inks, glasses, and paints and as a colorant for ceramics, as well as producing a faint green tinge in glazes [8].
- It serves as an abrasive for polishing or stropping the edges of razors, knives, and surfaces of optical devices [9].
Is It Safe
Exposure to chromium (iii) oxide through mouth and skin can cause acute toxicity [1]. It may cause an allergic skin reaction and serious eye damage [1]. It can also cause reproductive toxicity and is harmful to the unborn child [1].
- References
- Chromium(III) Oxide – Pubchem.ncbi.nlm.nih.gov
- Chromium Oxide Powder (Cr2O3) – Reade.com
- Chromium(III) Oxide – Americanelements.com
- Chemistry of Chromium – Chem.libretexts.org
- Chromium(III) Oxide – Chemspider.com
- Chemical Equations – Chemequations.com
- Stoichiometry of Ammonium Dichromate Decomposition – Dartmouth.edu
- Chromium Oxide – Chromestarchemicals.com
- 5 Interesting Facts About Green Chrome Oxide – Basstechintl.com
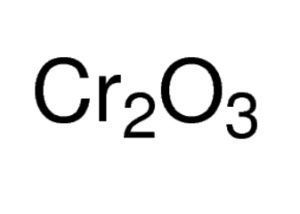






i would like to buy green chrome oxide for refractory use. use as a non-wetting agent and making my castable basic