Disproportionation Reaction
What is Disproportionation Reaction [1-3]
A disproportionation reaction is a redox reaction in which an element present in a particular compound is oxidized and reduced simultaneously. It is considered a redox reaction because oxidation and reduction occur in the same reaction. To find a disproportionation reaction, always look for an element having at least three oxidation states in the reaction.
The reverse of the disproportionation reaction is called comproportionation or synproportionation. Here, two reactants containing a common element but a different oxidation number combine to yield a product. The element reaches an intermediate oxidation number.
Example
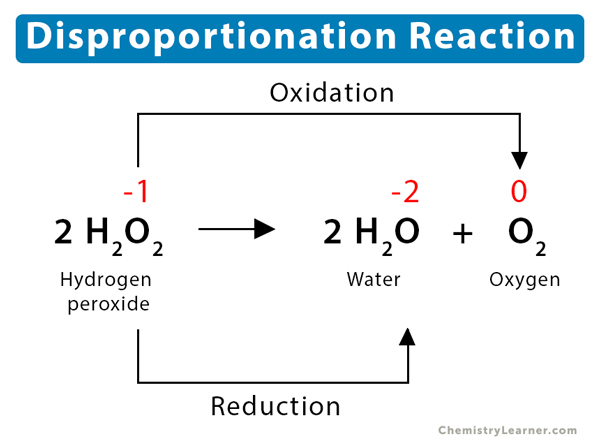
A prevalent example of a disproportionation reaction is the decomposition of hydrogen peroxide. Upon dissociation, the relatively less stable H2O2 forms more stable H2O and O2.
2H2O2 → 2H2O + O2
The oxidation state of the oxygen atom in H2O2 is -1. This oxygen atom gets reduced to H2O, where its oxidation state becomes -2. The same oxygen atom also gets oxidized to O2, gaining the oxidation state 0. The oxygen atom on the reactant is getting both oxidized and reduced simultaneously.
Other Examples of Disproportionation Reaction [1-3]
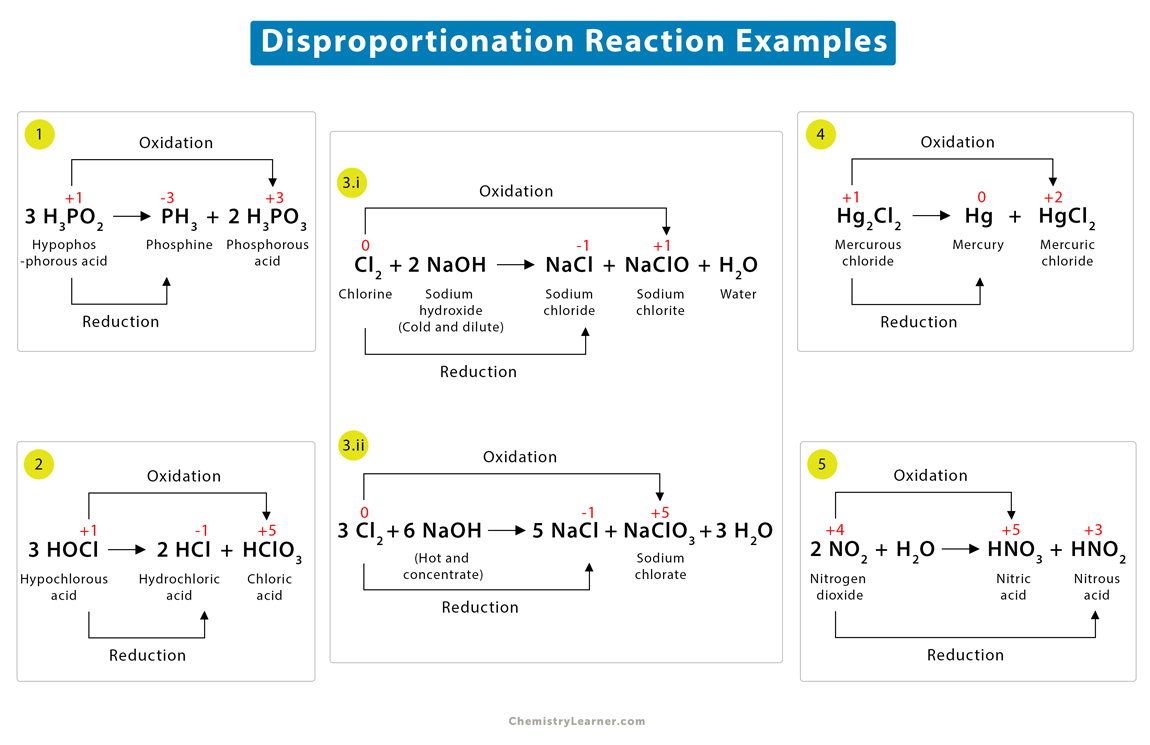
1. Dissociation of hypophosphorous acid (H3PO2)
H3PO2 gets decomposed into colorless phosphine gas (PH3) and phosphorous acid (H3PO3).
3H3PO2 → PH3 + 2H3PO3
Phosphorus atom is in +1 oxidation state in H3PO2. This phosphorus gets oxidized to H3PO3, where its oxidation state increases from +1 to +3. On the other hand, the same phosphorus atom gets reduced to PH3. Here, its oxidation state decreases from +1 to -3.
2. Decomposition of hypochlorous acid (HOCl)
HOCl dissociates into hydrochloric acid (HCl) and chloric acid (HClO3).
3HOCl → 2HCl + HClO3
The oxidation state of the chlorine atom in HOCl is +1, which gets oxidized to HClO3, reaching the oxidation state of +5. The same chlorine atom also gets reduced to HCl, leading to an oxidation state -1.
3. Disproportionation reaction of chlorine (Cl2)
i) Chlorine reacts with a cold and dilute solution of sodium hydroxide (NaOH) to form sodium chloride (NaCl), sodium chlorite (NaClO), and water (H2O).
Cl2 + 2NaOH → NaCl + NaClO + H2O
On the reactant side, the chlorine atom has an oxidation state of 0. It gets oxidized to +1 in NaClO and reduced to -1 in NaCl.
ii) If chlorine gas is passed through a hot and concentrated solution of sodium hydroxide (NaOH), it is oxidized to sodium chlorate (NaClO3) instead of NaClO.
3Cl2 + 6NaOH → 5NaCl + NaClO3 + 3H2O
Here, the chlorine atom with oxidation state 0 reaches +5 in NaClO3 and -1 in NaCl.
4. Photolysis of mercurous chloride (Hg2Cl2)
Hg2Cl2 undergoes disproportionation under UV light to give mercury (Hg) and mercuric chloride (HgCl2).
Hg2Cl2 → Hg + HgCl2
The oxidation state of mercury in Hg2Cl2 is +1. It gets oxidized to HgCl2, reaching the oxidation state +2. It also gets reduced to Hg, changing from +1 to 0.
5. Reaction of nitrogen dioxide (NO2) with water
NO2 reacts with water to give nitric (HNO3) and nitrous (HNO2) acids.
2NO2 + H2O → HNO3 + HNO2
The nitrogen in NO2 is in a +4 oxidation state. It is oxidized to HNO3, in which the oxidation state of nitrogen is +5. It also gets reduced to HNO2, reaching the oxidation state +3.
How to Find n-factor in Disproportionation Reaction
The n-factor of a compound in a reaction is a number that measures the number of electrons lost or gained by it during oxidation and reduction per mole of the given compound. In a balanced disproportionation reaction, it can be observed that the numbers of electrons being gained or lost as a result of oxidation or reduction are the same.
To understand how to calculate the n-factor for a disproportionation reaction, let us take the example of dissociation of hypophosphorous acid (H3PO2) from the above section.
Phosphorus atom is in +1 oxidation state in H3PO2. This phosphorus gets oxidized to H3PO3, where its oxidation state increases from +1 to +3. On the other hand, the same phosphorus atom gets reduced to PH3. Here, its oxidation state decreases from +1 to -3.
Steps:
1. Assign the oxidation state to phosphorus atoms in each reactant and product. The phosphorus atom is in a +1 oxidation state on the reactant side. On the product side, the phosphorus is in +3 and -3 oxidation states, in H3PO3 and PH3, respectively.
2. Next, we need to break this disproportionation reaction into two halves – oxidation and reduction. Each half-reaction must be written, showing the number of electrons involved in the process.
i. Oxidation: H3PO2 → H3PO3 + 2e−
ii. Reduction: H3PO2 + 4e− → PH3
3. Note that the electrons in both the half-reactions are not balanced. To balance them, the oxidation half needs to be multiplied by 2.
2H3PO2 → 2H3PO3 + 4e−
Therefore, 4 electrons and 3 molecules are involved in this disproportionation reaction.
Calculating the n-factor
The formula for n−factor calculation is given as follows:
n−factor = (number of electrons involved) / (number of molecules involved)
Putting the values,
Therefore, n−factor = 4/3
References
- Disproportionation: Definition & Examples – Study.com
- Disproportionation Definition in Chemistry – Thoughtco.com
- Oxidation States & Redox Reactions – Chem.libretexts.org