Livermorium
What is Livermorium
Livermorium (pronounced as liv-er-MORE-ee-em [2]) is a synthetic, radioactive metal [3] denoted by the chemical symbol Lv [1]. Four isotopes of the element with their mass numbers between 290 and 293 have been produced so far. Of them, the most stable one is Lv 293 with a half-life of 61 milliseconds [1]. It belongs to the family of post-transition elements [9].
Where is it Found
It is not found naturally on the earth. Small amounts of the metal have been produced in the lab [1].
History
Origin of Its name: It is named after the Lawrence Livermore National Laboratory (LLNL) in California, USA [1].
Who Discovered It: A team of scientists led by Yuri Oganessian, Vladimir Utyonkov and Kenton Moody at the Joint Institute of Nuclear Research (JINR) in Dubna, Russia [1].
How Was It Discovered
There were several attempts to make the element, all of which were unsuccessful until 2000 when researchers at JINR synthesized it for the first time. Since the discovery utilized target material supplied by LLNL, the element was named after the facility. It was (is) created in the laboratory by bombarding curium atoms with those of calcium [1].
Livermorium Identification |
|||
| Atomic Number | 116 [1] | ||
| CAS Number | 54100-71-9 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 16 [1] | 7 [1] | p [1] | |
Properties and Characteristics of Livermorium
General Properties |
||
| Atomic mass | 293 atomic mass units [1] | |
Physical Properties |
||
| Color/appearance | Unknown [10] | |
| Melting point/freezing point | Unknown [1] | |
| Boiling point | Unknown [1] | |
| Density | Unknown [1] | |
| State of matter at room temperature (normal phase) | Solid (presumably) [1, 2, 3] | |
Chemical Properties |
||
| Oxidation state/Oxidation number | [+2], [+4] [8] | |
| Reactivity | Unknown [8] | |
Atomic Data of Livermorium (Element 116)
| Valence electrons | 6 [6] | ||||||
| Quantum numbers | |||||||
| – n | 7 [6] | ||||||
| – ℓ | 1 [6] | ||||||
| – m ℓ | -1 [6] | ||||||
| – m s | -½ [6] | ||||||
| Electron configuration (noble gas configuration) | [Rn] 5f146d107s27p4 [1] | ||||||
| Atomic structure | |||||||
| – Number of Electrons | 116 [3] | ||||||
| – Number of Neutrons | 177 [3] | ||||||
| – Number of Protons | 116 [3] | ||||||
| Energy levels [3] | |||||||
| – First Energy Level | 2 | ||||||
| – Second Energy Level | 8 | ||||||
| – Third Energy Level | 18 | ||||||
| – Fourth Energy Level | 32 | ||||||
| – Fifth Energy Level | 32 | ||||||
| – Sixth Energy Level | 18 | ||||||
| – Seventh Energy Level | 6 | ||||||
| Radius of atom | |||||||
| – Atomic Radius | Unknown [1] | ||||||
| – Covalent Radius | 1.75 Å [1] | ||||||
| Electronegativity | Unknown [7] | ||||||
| Ionization energy
(kJmol-1) [1] |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| – | – | – | – | – | – | – | |
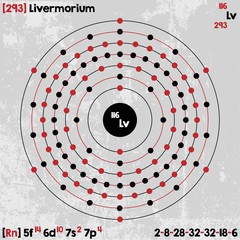
Livermorium Electron Configuration (Bohr Model)
Uses of Livermorium
Since only a few atoms of the element have been made to date, its use is limited to laboratory research [1].
Can it be Dangerous
The naturally radioactive property of the element makes it potentially harmful [3].
Interesting Facts
- Both livermorium and flerovium were created by a collaboration between the JINR, Russia and LLNL, USA [1] and they were both given their official names by IUPAC in May 2012 [4, 5] when they were added to the periodic table [12].
- It was given the temporary name ununhexium (symbol Uuh [13]), denoting its atomic number 116, before getting its official name [3].
- The city of Livermore celebrates The Livermorium Day on May 30 every year to honor the discovery of the element named after it [11].
Livermorium (Ununhexium) Cost
It is not available commercially and is only produced in laboratories in minuscule amounts [1, 3].
- References
- http://www.rsc.org/periodic-table/element/116/livermorium
- https://education.jlab.org/itselemental/ele116.html
- https://www.chemicool.com/elements/livermorium.html
- https://www.livescience.com/17287-element-names-flerovium-livermorium.html
- https://phys.org/news/2012-05-livermorium-flerovium-periodic-table-elements.html
- http://chemistry-reference.com/q_elements.asp?Symbol=Lv&language=en
- https://www.webelements.com/livermorium/electronegativity.html
- https://www.webelements.com/livermorium/chemistry.html
- https://www.elements-table.com/elements/livermorium/
- https://www.americanelements.com/livermorium.html
- http://www.independentnews.com/livermorium-day-celebration/collection_25dc0934-ed93-11e3-ae8f-001a4bcf887a.html
- https://www.llnl.gov/news/livermorium-and-flerovium-join-periodic-table-elements
- https://www.radiochemistry.org/periodictable/elements/116.html