Oxidation Number (Oxidation State)
What is Oxidation Number
An oxidation number is a number that is assigned to an atom to indicate its state of oxidation or reduction during a chemical reaction. Each atom in a redox reaction is assigned an oxidation number to understand its ability to donate, accept, or share electrons. It shows the total number of electrons that have been removed from or added to an element to get to its present state. For example, in Fe2O3, the oxidation number of Fe is +3, and in FeO, it is +2. A loss of electrons corresponds to an increase in oxidation number. On the other hand, a gain of electrons corresponds to a decrease in oxidation number [1-4].
Oxidation Number Rules [1-6]
In order to assign oxidation numbers to atoms, we need to follow a set of rules.
1. The oxidation number of an element in its free state is zero.
Example: The oxidation number of Zn, Al, H2, O2, and Cl2 is zero
2. The oxidation number of a monatomic ion is the same as the charge on the ion.
Example: The oxidation number of Na+ is +1, Mg2+ is +2, Al3+ is +3, Cl-1 is -1, and O2- is -2.
3. The sum of all oxidation numbers in a neutral compound is zero. The sum of all oxidation numbers in a polyatomic ion is equal to the charge on the ion.
Example: In Fe2O3, the oxidation number of Fe is +3, and that of O is -2. The sum of all oxidation numbers is: +3 x 2 + (-2) x 3 = 0. The result is expected since Fe2O3 is neutral.
4. The oxidation number of an alkali metal in a compound is +1, and the oxidation number of an alkaline earth metal in a compound is +2.
Example: In NaCl, Na is an alkali metal, and its oxidation number is +1. In MgO, Mg is an alkaline earth metal, and its oxidation number is +2.
5. The oxidation number of oxygen in a compound is usually -2. However, if the oxygen is in a category of compounds called peroxides, its oxidation number is -1. If the oxygen is bonded to fluorine, the number is +1 or +2, depending upon the compound.
Example: The oxidation number of O in H2O is -2, in H2O2 is -1, in OF2 is +2, and in O2F2 is +1
6. The oxidation number of hydrogen in a compound is usually +1. In the case of a binary metal hydride, the oxidation number is -1.
Example: The oxidation number of H is +1 in H2O and -1 in NaH.
7. The oxidation number of fluorine is always -1.
Example: The oxidation number of F in NaF is -1.
8. Chlorine, bromine, and iodine usually have an oxidation number of -1 unless bonded to oxygen or fluorine.
Example: The oxidation number of Cl in NaCl is -1 and in ClO2 is +4, and in FCl is +1.
How to Find Oxidation Number
The oxidation number of an atom in an ion or compound can be determined using the above rules. Let us look at a few examples [1-6].
1. Sulfuric Acid (H2SO4)
The oxidation number of hydrogen (H) and oxygen (O) are +1 and -2, respectively. Sulfuric acid is a neutral compound. Let x be the oxidation number of sulfur (S). Therefore,
(+1) x 2 + x + (-2) x 4 = 0
Or, 2 + x – 8 = 0
Or, x = +6
2. Nitric Acid (HNO3)
The oxidation numbers of hydrogen (H) and oxygen (O) are +1 and -2, respectively. Nitric acid is a neutral compound. Let x be the oxidation number of nitrogen (N). Therefore,
+1 + x + (-2) x 3 = 0
Or, +1 + x – 6 = 0
Or, x = +5
3. Potassium Permanganate (KMnO4)
The oxidation numbers of potassium (K) is +1 and oxygen (O) is -2. KMnO4 is a neutral compound. Let x be the oxidation number of magnesium (Mn). Therefore,
+1 + x + (-2) x 4 = 0
Or, +1 + x – 8 = 0
Or, x = +7
4. Dichromate Ion (Cr2O72-)
Dichromate is a complex ion. The oxidation number of oxygen (O) is -2. The charge of Cr2O72- is -2. Let x be the oxidation number of chromium (Cr).
2x + (-2) x 7 = -2
Or, 2x -14 = -2
Or, x = +6
5. Carbonate (CO32-)
The oxidation number of oxygen (O) is -2 and the charge on CO32- is -2. Let x be the oxidation number of carbon (O). Therefore,
x + (-2) x 3 = -2
Or, x – 6 = -2
Or, x = +4
6. Phosphite (PO33-)
The oxidation number of oxygen (O) is -2 and the charge of PO33- is -3. Let x be the oxidation number of phosphorous (P). Therefore,
x + (-2) x 3 = -3
Or, x – 6 = -3
Or, x = +3
7. Potassium Perchlorate (KClO4)
The oxidation numbers of potassium (K) and oxygen (O) are +1 and -2, respectively. Let x be the oxidation number of chlorine (Cl). KClO4 is a neutral compound. Therefore,
+1 + x + (-2) x 4 = 0
Or, 1 + x – 8 = 0
Or, x = +7
8. Potassium Nitrate (KNO3)
The oxidation number of potassium (K) and oxygen (O) are +1 and -2, respectively. KNO3 is a neutral compound. Let x be the oxidation number of nitrogen (N). Therefore,
+1 + x + (-2) x 3 = 0
Or, 1 + x – 6 = 0
Or, x = +5
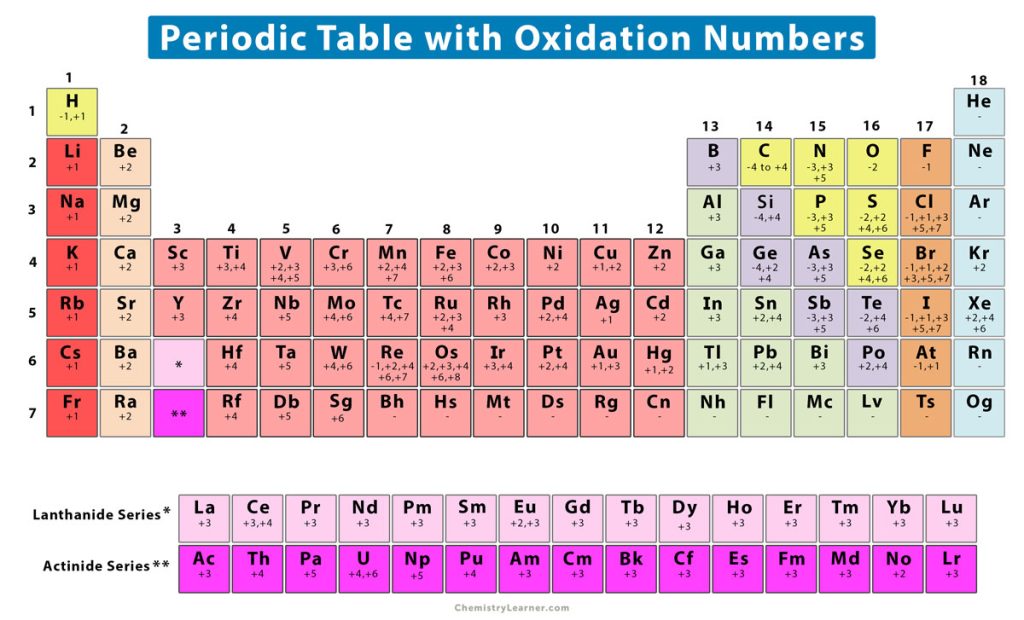
The following image shows a chart consisting of the oxidation numbers of the periodic table elements [7].
FAQs
Ans. The difference between valency and oxidation number is that valency is the maximum number of electrons an atom can donate, accept, or share to become stable. In contrast, the oxidation number is the number of electrons an atom can donate or accept to form a bond with another atom.
Ans. The d-block or translational elements have incomplete d- and s-subshells. The valence electrons are present in both these subshells. It is for this reason that they can form variable oxidation states.