Radon
What is Radon
Radon (pronunciation: RAY-don) is an odorless, radioactive element that belongs to the family of noble gases and is represented by the chemical symbol Rn [1, 2]. It is colorless at room temperatures, but becomes phosphorescent when cooled below its freezing temperature and gradually turns into orange-red when the temperature is further lowered to the boiling point of liquid air [4].
Isotopes
There are more than 35 radioactive isotopes of radon, ranging from 195Rn-229Rn, out of which 222Rn is the most stable with a half-life period of 3.823 days and forms 218Po through α-decay [5]. It has four naturally occurring isotopes, including 218Rn, 219Rn, 220Rn, and 222Rn [5].
Where is Radon Found
Radon is always found in nature because it is produced from the radioactive decomposition of radium-226 that is present in phosphate rocks, uranium ores, metamorphic rocks, and common rocks like limestone [1, 3]. A small amount of radon also occurs in the atmosphere [1].
History
Origin of its Name: It is named for the radioactive metallic element radium because radon was first identified during the radioactive decay of radium [1].
Who discovered it: The German physicist Friedrich Ernst Dorn is known for its discovery [1].
When and How was it Discovered
In 1899, Robert B. Owens and Ernest Rutherford found a radioactive gas emanating from thorium [1]. In that same year, Marie and Pierre Curie detected a gas being released by radium [1]. In 1900, Ernst Dorn discovered a gas while studying the decay chain of radium at the German city of Halle [1].
We now know that the radioactive gas observed by the Curies and Ernst Dorn was Radon-222 while the gas detected by Rutherford was Radon-220 [1]. In 1908, Robert Whytlaw-Gray and William Ramsay collected enough radon and determined the properties of radon at the University College, London [1].
Radon Identification |
|||
| Atomic number | 86 [1] | ||
| CAS number | 10043-92-2 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 18 [1] | 6 [1] | p [1] | |
Properties and Characteristics of Radon
General Properties |
||||||||||||||||
| Relative atomic mass | [222] [1] | |||||||||||||||
| Atomic mass | [222] amu [1] | |||||||||||||||
| Molar mass | 210.9906010 ± 0.0000070 g/mol [6] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Colorless [1, 7] | |||||||||||||||
| Melting point/freezing point | -71 °C, -96 °F [1] | |||||||||||||||
| Boiling point | -61.7 °C, -79.1 °F [1] | |||||||||||||||
| Density | 0.009074 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Gas [1, 7] | |||||||||||||||
| Charge | Unknown [8] | |||||||||||||||
| Thermal (heat) conductivity | 0.00361 W/(m K) [9] | |||||||||||||||
| Flammability | Not flammable [10] | |||||||||||||||
| Specific heat | 94 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | Unknown [1] | |||||||||||||||
| Shear modulus | Unknown [1] | |||||||||||||||
| Young’s modulus | Unknown [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | – | – | – | – | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | +2, (+6)[1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 211Rn | 210.991 | – | 14.6 h | β+, EC | ||||||||||||
| – | α | |||||||||||||||
| 220Rn | 220.011 | – | 55.6 s | α | ||||||||||||
| 222Rn | 128.905 | – | 3.823 d | α | ||||||||||||
Atomic Data of Radon (Element 86)
| Valence electrons | 8 [11] | |||||||
| Quantum numbers | ||||||||
| – n | 6 [11] | |||||||
| – ℓ | 1 [11] | |||||||
| – mℓ | 1 [11] | |||||||
| – ms | -1/2 [11] | |||||||
| Electron configuration (noble gas configuration) | [Xe] 4f145d106s26p6 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 86 [7] | |||||||
| – Number of neutrons | 136 [7] | |||||||
| – Number of protons | 86 [7] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.20 Å [1] | |||||||
| – Covalent radius | 1.46 Å [1] | |||||||
| Electronegativity (Pauling-scale) | Unknown [1] | |||||||
| Electron affinity | Not stable [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 1037.073 | – | – | – | – | – | – | – | |
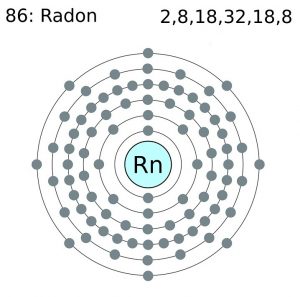
Radon Electron Configuration (Bohr Model)
What is it Used for
- Radioactive decomposition of radon is useful for obtaining polonium [1].
- Radon emitted from a radium source can be used in cancer therapy [1]. It was commonly used in hospitals for the treatment of tumors through implantation of minute tubes in which the gas was sealed [1, 7]. Today, it has been replaced by safer treatment options [1, 7].
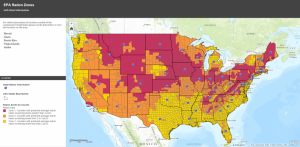
- Radon testing kits are used for analyzing indoor radon levels in places where large concentrations of radon gas can accumulate indoors [1]. If the test result shows the level is 4 pCi/L or more, mitigation systems are used to reduce radon concentrations [12].
- Researchers use radon soil-concentration to map subsurface geological faults since concentrations generally increase over the faults [13]. Groundwater radon concentrations are also analyzed for earthquake prediction [14].
Radon Toxicity and Health Effects
The decay products of radon have been considered as being carcinogenic [7]. Since it can be inhaled, people exposed to high levels of radon are at risk of developing lung cancer [15]. The chances of getting affected by lung cancer are much higher in smokers who are exposed to radon [15]. It is thought to have had played a role in evolution and could be responsible for the background radiation of the Earth which can cause genetic modifications [1].
Interesting Facts
- It is the heaviest known chemically inactive gas that can form compounds with other substances only under extreme conditions [3, 4].
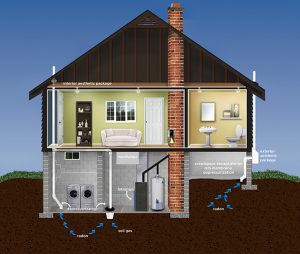
- It is graphically represented by an image of the radiation hazard symbol with background images of houses indicating that the gas can accumulate in houses [1].
Radon Cost
The price of radon gas is about $4 per m [16].
- References
- http://www.rsc.org/periodic-table/element/86/radon
- https://education.jlab.org/itselemental/ele086.html
- http://www.chemistryexplained.com/elements/P-T/Radon.html
- https://www.livescience.com/39546-radon.html
- https://education.jlab.org/itselemental/iso086.html
- https://www.webqc.org/molecular-weight-of-radon.html
- https://www.chemicool.com/elements/radon.html
- Lenntech.com
- http://periodictable.com/Elements/086/data.html
- https://www.atsdr.cdc.gov/toxprofiles/tp145-c4.pdf
- http://chemistry-reference.com/q_elements.asp?Symbol=Rn&language=en
- https://sosradon.org/mitigation
- Sciencedirect.com
- https://www.jstage.jst.go.jp/article/jpe1952/43/5/43_5_585/_pdf
- https://www.epa.gov/radon/health-risk-radon
- https://hobart.k12.in.us/ksms/PeriodicTable/radon.htm