Swern Oxidation
Definition: What is Swern Oxidation?
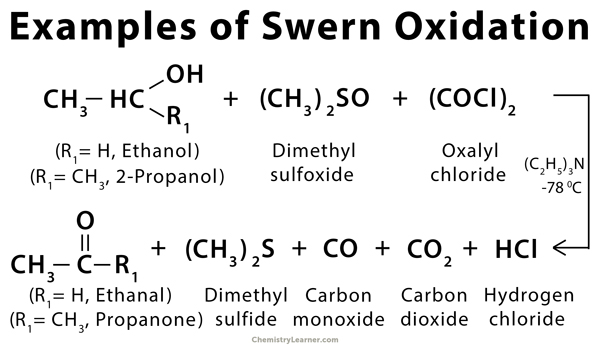
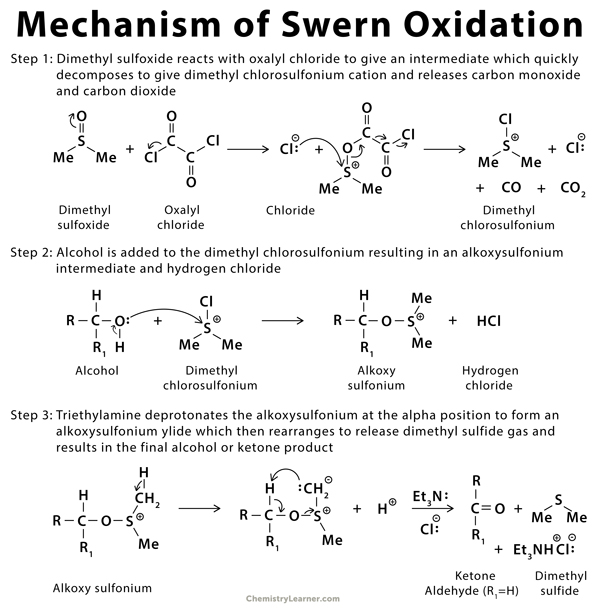
Swern oxidation is an organic chemical reaction that is used to oxidize primary or secondary alcohol into an aldehyde or ketone. The reagents used in this reaction are oxalyl chloride, dimethyl sulfoxide (DMSO), and an organic base like triethylamine [1-4].
Examples of Swern Oxidation
Mechanism of Swern Oxidation [3-7]
Conditions of Swern Oxidation
The oxidation reaction should be carried out under very mild conditions and below 60 °C to avoid side reactions [1].
Applications of Swern Oxidation
The reaction is used for large scale synthesis of aldehydes because the byproducts derived from the reagents are low boiling and easy to remove [8].
FAQs
Q.1. What precautions should be taken for dimethyl sulfide?
Ans. Dimethyl sulfide, which is a byproduct, is a foul-smelling chemical and must be removed immediately.
Q.2. What are the alternatives to Swern oxidation?
Ans. Alternatives to Swern oxidation can include other reagents for the activation of DMSO. Dicyclohexylcarbodiimide (DCC) (Pfitzner-Moffatt oxidation), trifluoroacetic anhydride (TFAA) (modified Swern oxidation), acetic anhydride (Albright-Goldmann oxidation), and SO3-pyridine (Parikh-Doering oxidation) are some of the alternative reagents.
Q.3. What is the advantage of using Swern oxidation over Jones oxidation?
Ans. The primary advantage of using Swern oxidation over Jones Oxidation is that the aldehyde formed from the oxidation of primary alcohol does not undergo further oxidation into a carboxylic acid.
- References
- Definition – Chem.libretexts.org
- Definition – Chem.libretexts.org
- Definition and mechanism – Byjus.com
- Definition and mechanism – Name-reaction.com
- Mechanism – Chemtube3d.com
- Mechanism – Organic-chemistry.org
- Mechanism – Ursula.chem.yale.edu
- Applications – En.chem-station.com