Coordinate Covalent Bond
What is a Coordinate Covalent Bond?
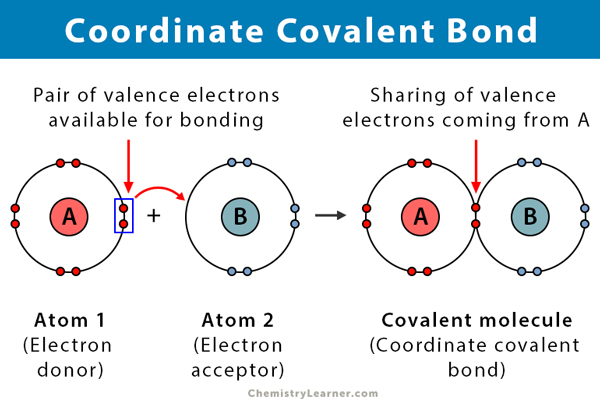
A covalent bond is formed when two atoms share electrons. A coordinate covalent bond, also known as a dative covalent bond, is a type of covalent bond in which the shared pair of electrons comes from one of the atoms. The atoms are held together because both of the nuclei attract the electron pair. Once the covalent bond is formed, it is impossible to distinguish the origin of the electrons. This kind of bond is typically observed in the bonding of metal ions to ligands. However, nonmetals can also participate in this bonding. The reaction between Lewis acid and base is a coordinate covalent bond [1-4].
A complex ion is made of two things, a metal ion and compounds called ligands. Ligands are neutral molecules or ions containing lone electron pairs that can bond with the metal ion. Common ligands are ammonia (NH3), water (H2O), and halide ions (Cl–, Br–). Ligands are considered Lewis bases because they are sharing their electron pairs with the metal ion. Metal ions are always positive, so they are quite attractive to lone pairs of electrons.
Facts and Properties of Coordinate Covalent Bond [5]
The coordinate covalent bond has many similarities to that of the covalent bond. Here are some of its properties:
- Takes place between an electron donor and an electron acceptor, typically between a metal and a ligand
- Takes place between dissimilar atoms
- Formed by the sharing of electrons donated by one of the atoms
- Rigid and directional
- Polar
- Atoms follow the octet rule
- The atom donating the electrons is called a donor, and the atom accepting them is called an acceptor.
- Compounds are sparingly soluble in water
Examples of Coordinate Covalent Bond
Here are some examples of molecules where a coordinate covalent bond exists [1-4].
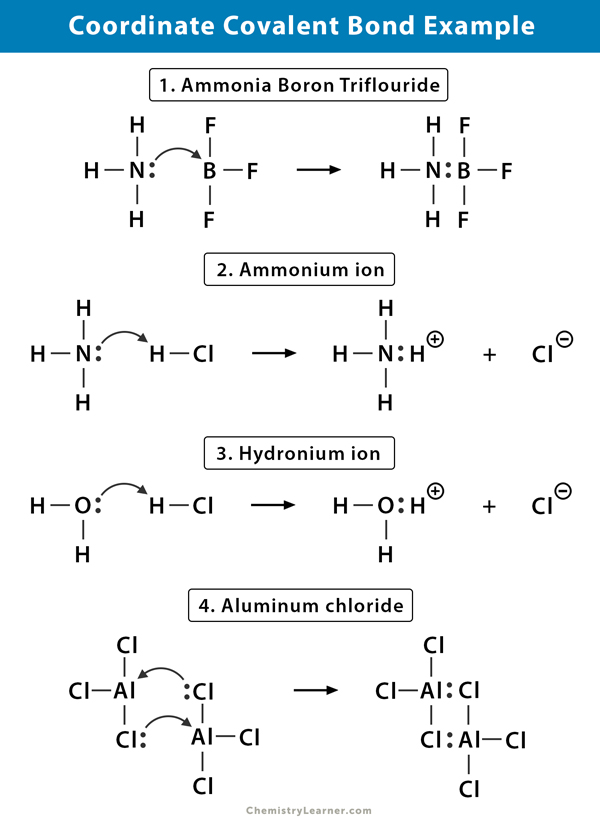
1. Ammonium (NH4+) ion
The ammonium ion is formed from the reaction of ammonia (NH3) gas with hydrogen chloride (HCl) gas. In NH4+, the fourth hydrogen is attached by a coordinate covalent bond because only the hydrogen’s nucleus is transferred from the chlorine to the nitrogen. The hydrogen’s electron is left behind on the chlorine to form a negative chloride (Cl–) ion.
2. Ammonia Boron Trifluoride (NH3.BF3)
Boron trifluoride (BF3) is a compound that does not have a noble gas structure around the boron (B) atom. The boron only has three pairs of electrons in its valence shell and requires a pair to complete the orbital. Hence, BF3 is electron deficient. The lone pair on the nitrogen (N) of the ammonia (NH3) molecule is used to overcome that deficiency, and a complex compound forms through a coordinate covalent bond.
3. Hydronium ion (H3O+)
When hydrogen chloride (HCl) gas dissolves in water to make hydrochloric acid (HCl aq.), a coordinate covalent bond is formed in the hydronium ion. The hydrogen (H) nucleus is transferred to the water (H2O) molecule, which has a lone pair of electrons to form hydronium. So, H does not contribute any electrons to the bond.
4. Aluminum Chloride (Al2Cl6)
The bonding in aluminum chloride (AlCl3) is essentially covalent. Each aluminum (Al) atom has a deficit of two electrons in its valence shell, and chlorine (Cl) has a lone pair. Al forms a coordinate covalent bond with the Cl atom on an adjacent AlCl3 group. As each of two Al atoms does this, then aluminum chloride is a covalent dimer molecule with the formula Al2Cl6.
5. Carbon monoxide (CO)
Carbon (C) has four electrons in its valence shell, and oxygen (O) has six. Both carbon and oxygen share their two electrons. While the octet rule is satisfied with oxygen, there is still a deficit of two electrons on the carbon. So, oxygen shares its two electrons with carbon to form a coordinate covalent bond, in addition to the two regular (double) covalent bonds.