Mass Spectrometry
Mass spectrometry is a powerful analytical technique for identifying and quantifying molecules based on their mass-to-charge ratio. It is a method to determine a sample’s chemical composition by measuring the mass of its individual components. [1-4]
Principle
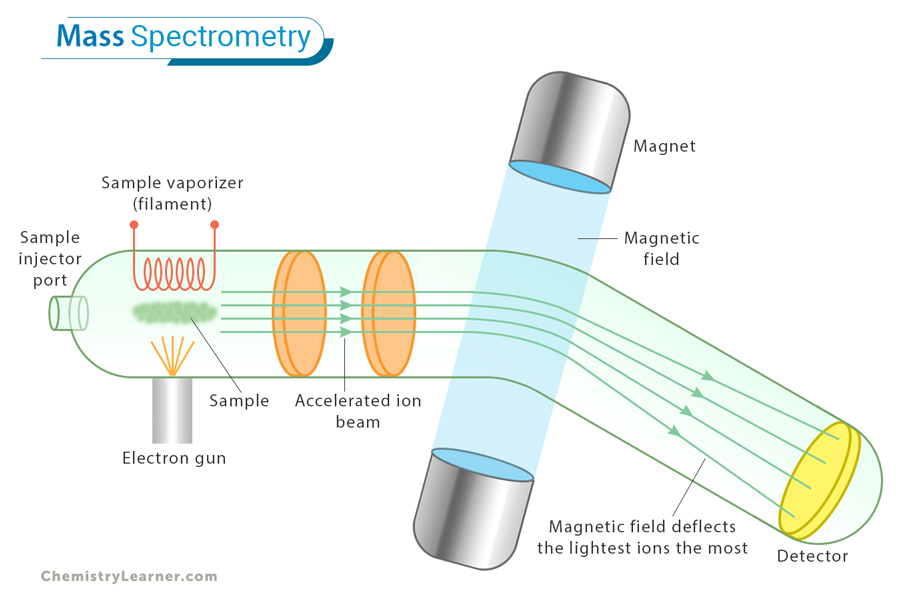
A mass spectrometer is the instrument used to perform mass spectrometry. It works by ionizing chemical compounds to generate charged particles (ions) and separate them. The resulting data provides valuable information about the molecular structure and composition of the sample. Here is a general overview of the key steps in a mass spectrometry experiment: [1-4]
1. Ionization: The sample is first ionized, meaning that atoms or molecules are converted into ions by gaining or losing electrons. It can be done using electron impact ionization or electrospray ionization.
2. Acceleration: The generated ions are then accelerated by an electric field, causing them to move towards a detector.
3. Deflection: In the presence of a magnetic or electric field, the ions are deflected based on their mass-to-charge ratio. Heavier ions are deflected less than lighter ions.
4. Detection: The ions are detected based on their mass-to-charge ratio, and the resulting data are recorded as a mass spectrum, showing the relative abundance of ions at different mass-to-charge ratios.
5. Data Analysis: The mass spectrum is analyzed to identify the types of atoms or molecules in the sample. Peaks in the spectrum represent different ions, and their positions provide information about the masses.
Detectors
Detectors play a crucial role in analyzing ions generated from the sample. Various types of detectors are used, each with unique capabilities and applications. [1]
1. Electron Multipliers: Electron multipliers are sensitive detectors commonly used in mass spectrometry for detecting low-energy electrons. These detectors amplify the ion current by generating a cascade of secondary electrons, increasing sensitivity.
2. Faraday Cups: Faraday cups are simple yet robust detectors that measure the total charge of ions reaching them. They are often used in high-resolution mass spectrometers due to their ability to quantify ion currents accurately.
3. Microchannel Plates: Microchannel plates are versatile detectors with high spatial and temporal resolution capabilities. They consist of tiny channels multiplying the incident ions through electron cascades, making them ideal for time-of-flight mass spectrometry.
4. Photomultiplier Tubes: Photomultiplier tubes are photon detectors commonly employed in mass spectrometry for detecting light emitted from ion interactions or secondary processes. They offer excellent sensitivity and fast response times, making them suitable for various analytical techniques.
How to Read Mass Spectrometry
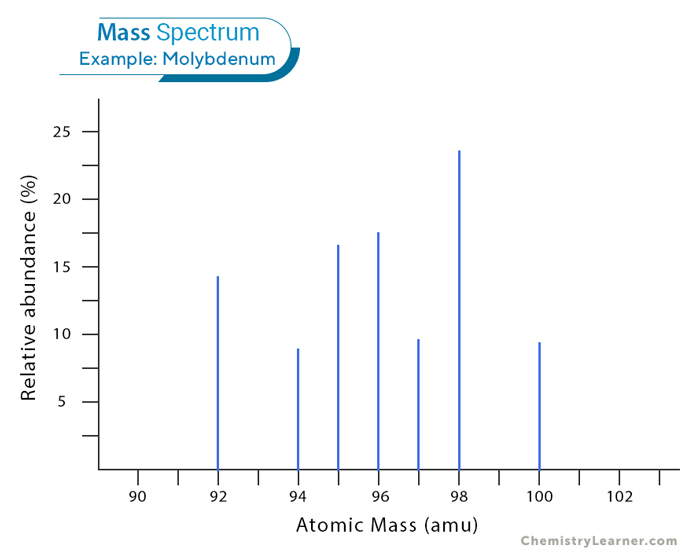
Mass spectrum provides a comprehensive snapshot of the isotopic composition of elements, offering valuable insights into the relative abundance and distribution of its isotopes based on their mass-to-charge ratios. Let us examine the mass spectrum of molybdenum. [1,3]
Examining the diagram reveals that the spectrum exhibits seven peaks, indicating the presence of seven naturally occurring isotopes of molybdenum. Notably, the height of each peak provides insight into the relative abundance of each molybdenum isotope compared to the others. The tallest peak represents the most abundant isotope. In the spectrum, the most prevalent ion in the sample exhibits a mass of 98. Additionally, the spectrum displays the presence of other ions with masses of 92, 94, 95, 96, 97, and 100.
This observation implies that molybdenum exhibits seven distinct isotopes. If we assume a 1+ charge for each ion, this information allows us to infer the average mass of molybdenum (95.94 amu).
Applications
Mass spectrometry is utilized in different industries that are discussed below: [1]
1. Pharmaceuticals: Mass spectrometry is extensively used in research and development. It aids in drug discovery by analyzing drug metabolites, assessing drug purity, and studying pharmacokinetics to ensure the safety and efficacy of medications.
2. Environmental Analysis: Environmental scientists rely on mass spectrometry to monitor pollutants in air, water, soil, and food samples. It enables them to identify contaminants, track their sources, and assess environmental impact accurately.
3. Forensic Science: Mass spectrometry is a valuable tool for identifying unknown substances found at crime scenes, detecting drugs or toxins in biological samples, and providing crucial evidence for criminal investigations.
4. Proteomics: Mass spectrometry is used to study proteins’ structure, function, and interactions within biological systems. It helps researchers gain insights into diseases like cancer and develop targeted therapies.