Periodic Trends
What are Periodic Trends
Periodic trends are specific patterns observed among the chemical elements of the periodic table. These observed patterns allude to the changes in atomic structure, including size and radius, as well as properties of the elements. These changes occur within their respective period from left to right and group from top to bottom in the periodic table [1-3,6].
Properties of Elements Based on Periodic Trends
1. Atomic Radius and Ionic Radius Trend
Atomic radius is the distance between an atom’s nucleus and its outermost or valence electrons. On the other hand, the ionic radius is half the distance between two ions that barely touch each other in a compound. The atomic and ionic radii follow the same trend in the periodic table. Hence, the discussion in this section will be of atomic radius [1-3,5].
Across a Period
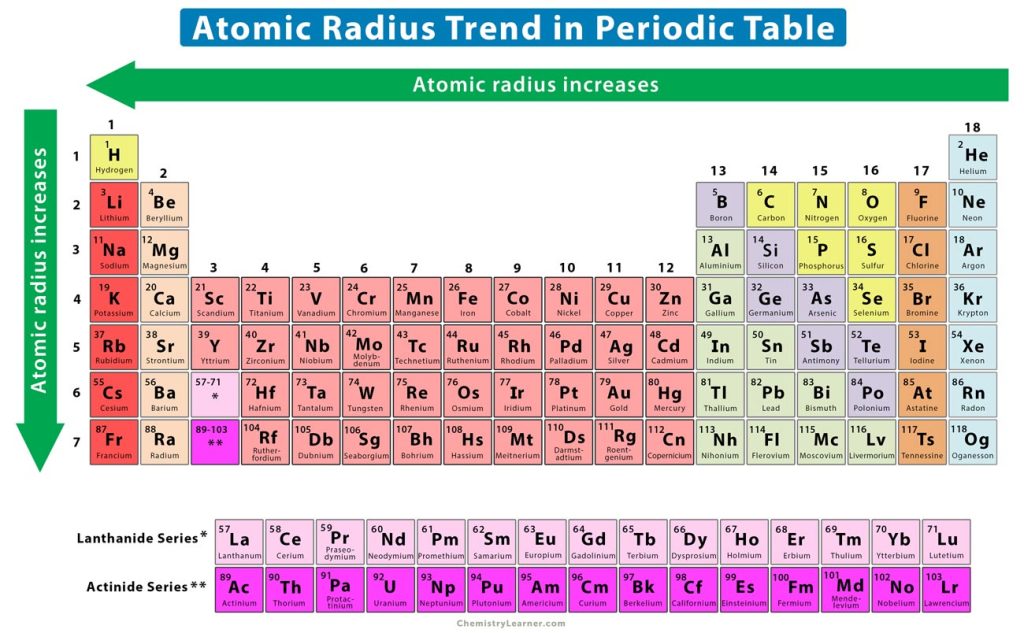
Along a period, electrons are added to the same shell of an atom as we go from left to right. Protons are also added to the atomic nucleus, making the nucleus more positively charged. As a result, the electrostatic attraction between the electrons and the nucleus increases, and the valence electrons are held closer to the nucleus. Thus, the atomic size and radius gradually decrease from left to right of a period. In other words, the atomic radius increases from right to left, as shown in the image below.
Down a Group
It is evident that as the atomic number increases down a group, the valence electrons occupy higher shells. The inner electrons shield the valence electrons and prevent them from getting closer to the nucleus. Hence, they are further away from the nucleus. Therefore, the atomic size and atomic radius increase from top to bottom.
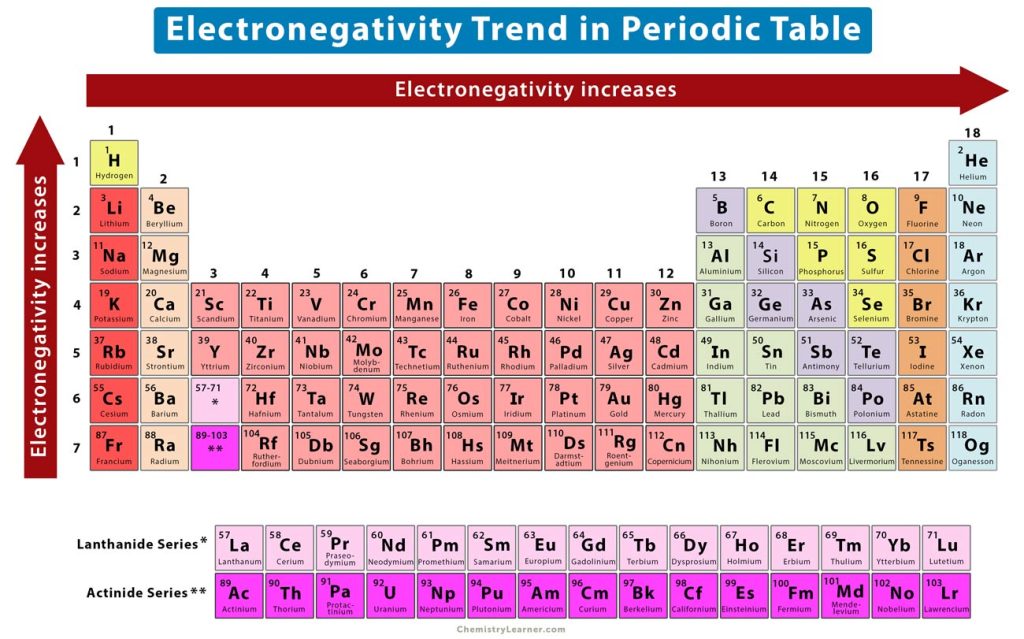
2. Electronegativity Trend
Electronegativity is the ability of an atom to attract electrons [1-3].
Across a Period
The atoms on the left of the periodic table have less than a half-full valence shell. They require more energy to attract electrons to complete their valence shell. As a result, they do not tend to attract electrons and have low electronegativity values. On the other hand, the atoms on the right have more than half-full valence shells and require less energy to acquire electrons to complete their valence shells. These atoms will have higher electronegativity values than the ones on the left. Therefore, the electronegativity increases from left to right across a period.
Down a Group
As mentioned before, the atomic size increases down a group. As a result, the electrostatic attraction between the nucleus and valence electrons decreases, making it difficult for the atoms to attract electrons. Therefore, the electronegativity decreases from top to bottom. In other words, the electronegativity increases from bottom to top, as shown in the image above.
Some exceptions to the above trend include noble gases, lanthanides, and actinides. The noble gases lie on the right of the periodic table, have a complete valence shell, and do not attract any electrons. Their electronegativity is zero. On the other hand, the lanthanides and actinides have complex atomic structures and do not follow any electronegativity trend.
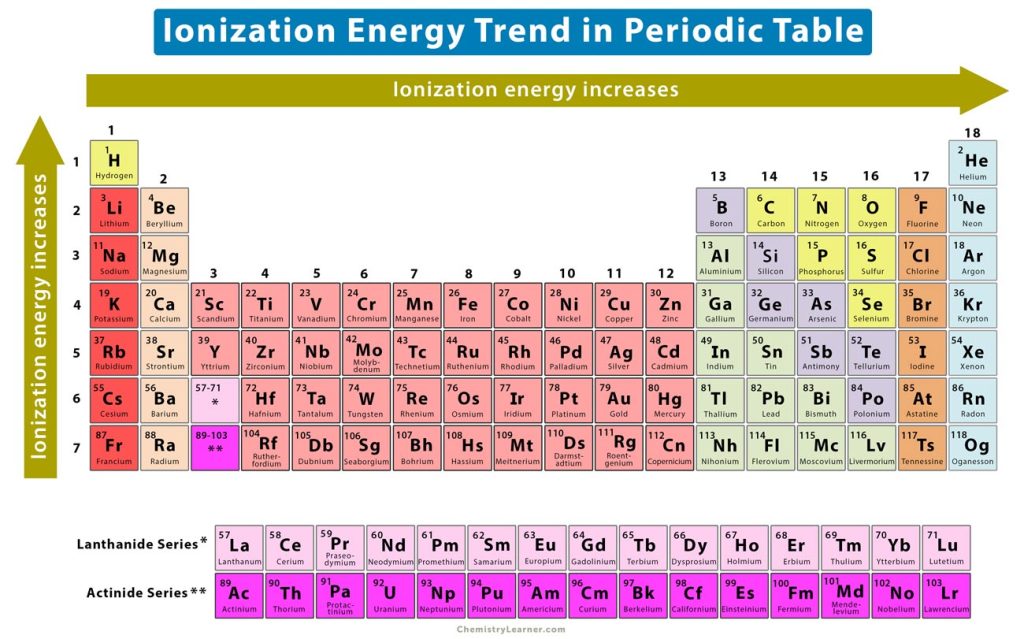
3. Ionization Energy Trend
Ionization energy is the minimum energy required to remove an electron from a neutral atom when the molecule is in the gaseous state. It is the opposite of electronegativity [1-4].
Across a Period
The elements on the right of the periodic table have nearly complete valence shells. Hence, it is not easy to remove an electron from them. These atoms will have higher ionization energies. On the other hand, the elements on the left have fewer electrons on the valence shell. They tend to lose electrons and take the configuration of their nearest inert gas elements. These atoms will have lower ionization energies. Thus, the ionization energy increases from left to right.
Down a Group
Down a group, the valence electrons are further away from the nucleus. It means that the electrostatic forces between the electrons and the nucleus are weak. Hence, the valence electrons are easy to remove. Thus, the ionization energy decreases from top to bottom. In other words, the ionization energy increases from bottom to top, as shown in the image above.
Effect of Shielding
There is another factor that affects ionization energy, which is the shielding of electrons. Shielding is defined as the ability of the inner electrons to shield the positively charged nucleus from outer electrons. The number of electrons increases down a group, so the shielding increases. The net nuclear charge experienced by a valence electron is known as the effective nuclear charge (Zeff). As shielding increases, the electric force between the nucleus and valence electrons reduces, making it easier to ionize the atom.
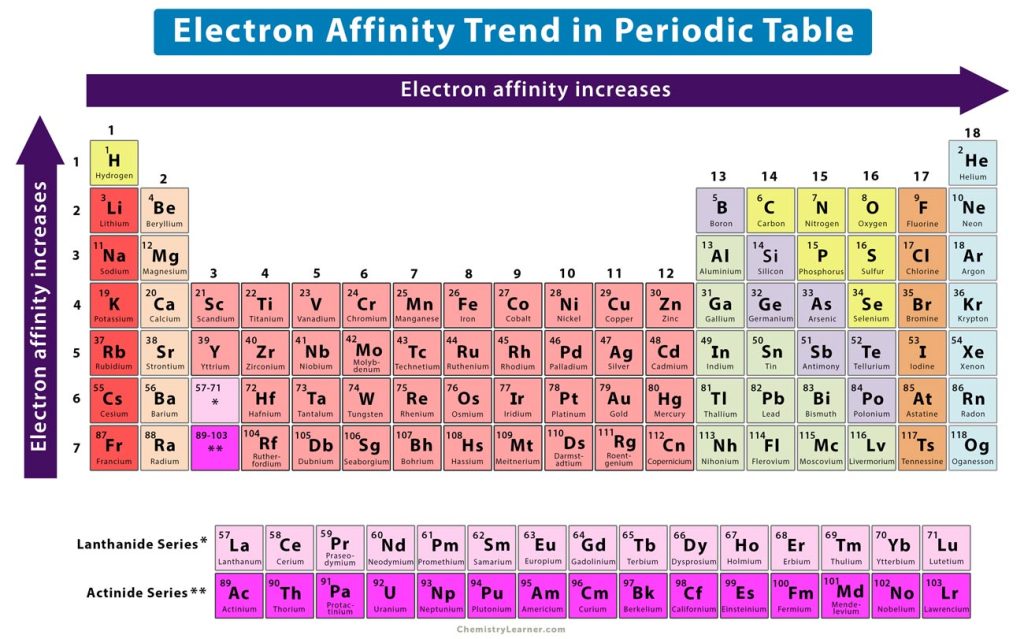
4. Electron Affinity Trend
The electron affinity is the change in energy when an electron is added to a neutral gaseous atom resulting in an anion. When an electron is added to an atom, it releases energy. Thus, the electron affinity takes a negative value. The more negative the electron affinity is, the more effortless adding the electron [1-3].
Across a Period
Across a period, the atoms become smaller due to the reason discussed in the section on atomic radius. So, when an electron is added to the valence shell, it will experience higher electrostatic attraction. The electron will move closer to the nucleus, thereby increasing the electron affinity. Thus, the electron affinity increases from left to right.
Down a Group
The atomic radius increases as the atomic number increases down a group. The increasing radius allows the electron to remain further from the nucleus. As this distance increases, the electrostatic force of attraction between the nucleus and electron becomes weaker. Thus, the electron affinity decreases from top to bottom. In other words, the electron affinity increases from bottom to top, as shown in the image above.
An exception to this trend is chlorine (period 3, group 17), which has a greater electron affinity than fluorine (period 2, group 17). The reason is that chlorine has more space for electrons in its outermost shell than fluorine. This larger space allows the chlorine atom to accommodate the extra electron, thus increasing the electron affinity.
5. Metallic and Non-metallic Character Trends
The metallic character of an element is the ability to lose an electron during a chemical reaction due to its low ionization energy. On the contrary, the non-metallic character is the ability to gain an electron during a reaction [1,3].
Across the Periods and Down the Groups
As discussed before, the elements in the bottom left of the periodic table have the lowest ionization energies. Hence, they are more reactive than other elements in their respective groups. They also have the lowest electron affinity. Thus, they are most metallic. Generally, the metallic character is displayed by the elements on the left of the periodic table. These elements are known as alkali and alkaline earth metals. The metallic character decreases across the periods from left to right and increases down the groups from top to bottom. An exception to this is hydrogen (H) which is a nonmetal.
On the other hand, the elements on the top right of the periodic table, except noble gases, have the highest ionization energy and electron affinity. Hence, they readily accept electrons during a chemical reaction. These elements are the least metallic. The non-metallic character trend is opposite to that of the metallic character.
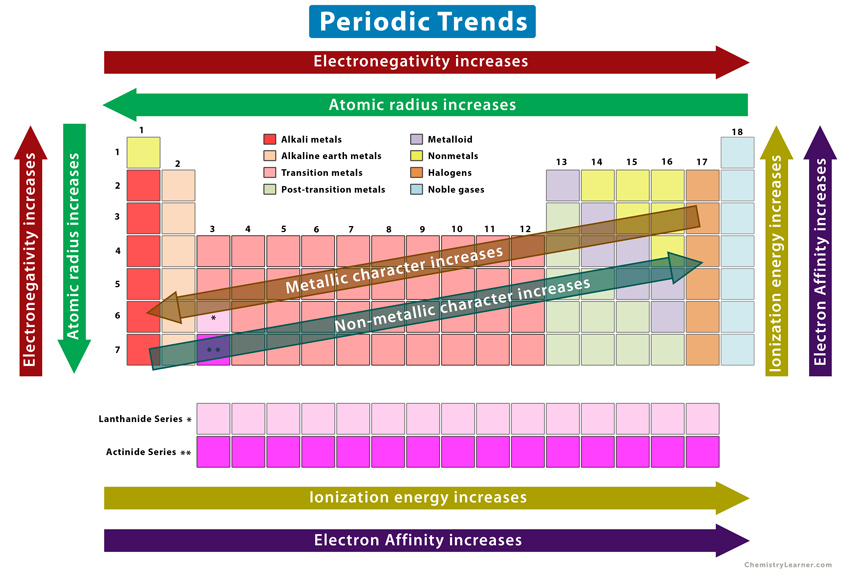
Summary
The following image summarizes all the periodic trends discussed in this article.
The elements on the left and center of the periodic table include alkali metals, alkaline earth metals, transition metals (including lanthanides and actinides), and post-transition metals. The elements on the right include nonmetals, halogens, and noble gases. In between lie the metalloids.
The electronegativity, ionization energy, and electron affinity increase from left to right and decrease from top to bottom. On the other hand, the atomic radius decreases from left to right and increases from top to bottom.
Starting from the bottom left and moving towards the top right, the metallic character decreases, and the non-metallic character increases. Therefore, alkali metals, alkaline earth metals, transition metals, and post-transition metals display metallic characteristics. The nonmetals, halogens, and noble gases display non-metallic characteristics. The metalloids display a mixture of both metallic and non-metallic behavior.