Coupling Reaction
What is Coupling Reaction
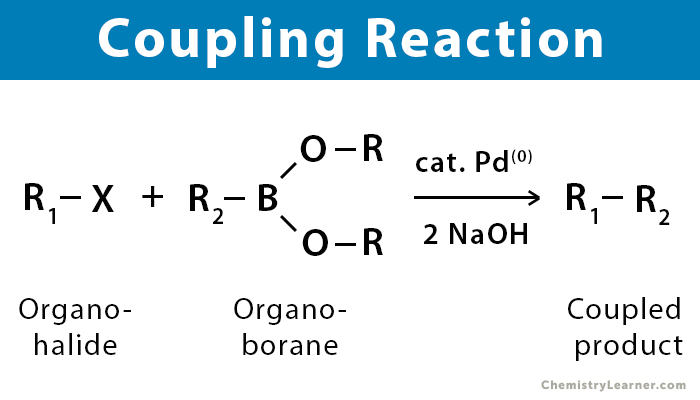
A coupling reaction in organic chemistry is a general term for various reactions where two fragments are joined together with the aid of a metal catalyst. Transition metal catalysts are used because they increase the reaction rate without affecting the heat of the reaction. During the reaction, transmetalation occurs in which an alkyl group is transferred from one metal to another. The energy is transferred from the reactants’ side to the product’s side [1-4].
History
American chemist Richard Heck did early pioneering work on palladium-catalyzed coupling reaction in the late 1960s.
What Happens in a Coupling Reaction
In a coupling reaction, an organometallic compound (R-M) reacts with an organic halide (R’-X) to form a new carbon-carbon bond in the product (R-R’).
Types and Examples of Coupling Reaction
There are two types of coupling reactions [3-4].
1. Homo-coupling Reaction: In this type of reaction, two identical species are joined together to form a new compound.
Examples: Wurtz reaction, Glaser coupling, and Pinacol coupling reaction
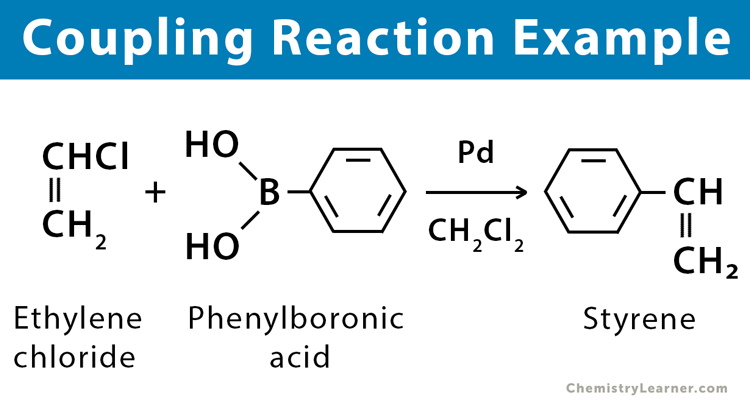
2. Hetero-coupling Reaction: In this type of reaction, two non-identical species are joined together to form a new compound. It is also known as a cross-coupling reaction and is common in arylation.
Examples: Heck reaction, Grignard reagent reaction, Suzuki reaction, Negishi coupling, and Hiyama coupling.
Applications of Coupling Reactions
The coupling reaction is used in various applications, including the synthesis of new and complex compounds like conjugated polymers and natural products. It is also used in the manufacture of new pharmaceutical drugs.
FAQs
Ans. The role of ATP in cells is to perform work by coupling the exergonic ATP hydrolysis’ reaction with endergonic reaction.
Ans. A diazo coupling reaction is the preparation of diazo compounds by coupling diazonium salts with a coupling reagent.