Dehydration Synthesis
What is Dehydration Synthesis?
Dehydration synthesis, also known as a dehydration reaction, is a chemical reaction that involves forming a new compound accompanied by removing water from the product. Some commonly used dehydrating agents are concentrated phosphoric acid, concentrated sulfuric acid, and alumina. When the synthesis occurs in the presence of heat, it is known as thermal dehydration [1-3].
Formula for Dehydration Synthesis
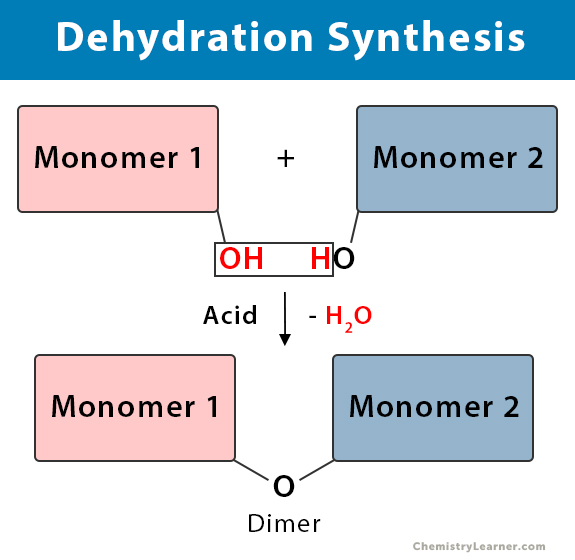
Monomer 1 – OH + HO – Monomer 2 → Monomer 1 – O – Monomer 2 + H2O
What Happens During Dehydration Synthesis?
Dehydration synthesis is a process in which two monomers combine covalently to form a dimer. One monomer loses a hydrogen (H) atom and the other a hydroxyl (OH) group. The H and OH combine to create a water (H2O) molecule. Water is a byproduct and eliminated from the reaction. Electrons play a key role indehydration synthesis. New covalent bonds are formed by sharing the electrons.
When many monomers combine in a series of steps, they form a polymer. This process is known as polymerization. Aside, amino acids combine to form peptides and proteins through dehydration synthesis. Simple carbohydrates like glucose also combine to synthesize a complex carbohydrate.
Examples of Dehydration Synthesis
The dehydration reaction is an essential reaction both in chemistry and biology. Here are some examples, along with their chemical equations [1-6].
1. Alcohol
Alcohols undergo dehydration reaction in the presence of a strong acid like sulfuric acid or phosphoric acid to yield alkenes.
Examples
- Dehydration of ethanol (C2H5OH) at 170 ⁰C gives ethene (C2H4)
CH3 – CH2 – OH → CH2 = CH2 + H2O
- Dehydration of 2-butanol (C4H9OH) gives 2-butene (C4H8)
CH3 – CH(OH) – CH2 – CH3 → CH3 – CH = CH – CH3 + H2O
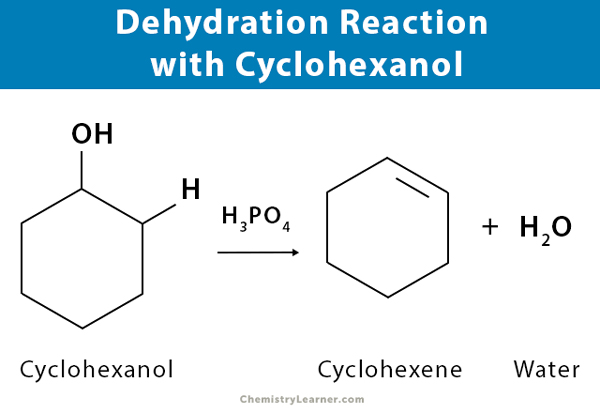
- Dehydration of cyclohexanol (C6H5OH) gives cyclohexene (C6H4)
C6H5 – OH → C6H4 + H2O
2. Carbohydrate
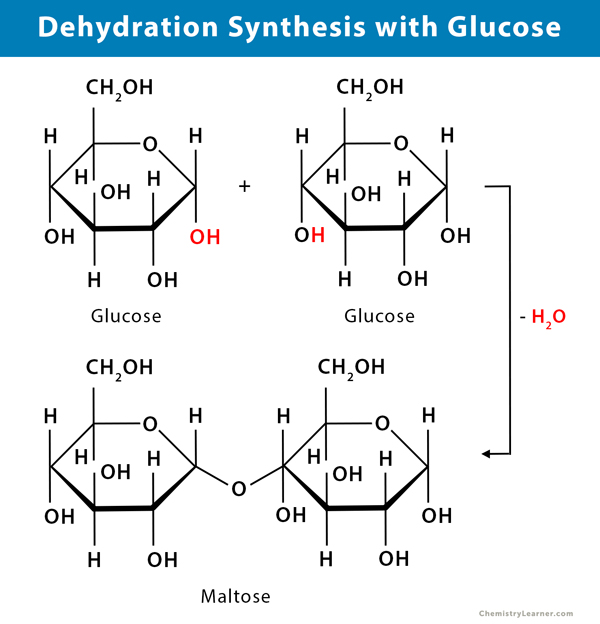
In the dehydration synthesis of carbohydrates like sugar, two monosaccharides combine to form a disaccharide.
Example
- Two molecules of glucose (C6H12O6) combine and undergo dehydration synthesis reaction to form a molecule of maltose (C12H22O11)
C6H12O6 + C6H12O6 → C12H22O11 + H2O
Likewise, lactose is formed by combining galactose and glucose, and sucrose by combining glucose and fructose.
3. Amino acid
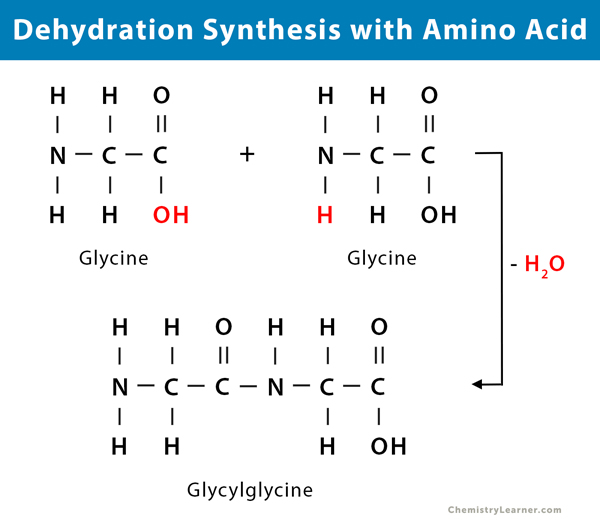
Two molecules of amino acid combine to form a peptide through the peptide bond linkage. This reaction is an example of a biological response. Peptides combine to form polypeptides.
Example
- Glycine condenses with itself to give a simple dipeptide called glycylglycine
NH2 – CH2 – COOH + NH2 – CH2 – COOH → NH2 – CH2 – CO – NH – CH2 – COOH + H2O
Fischer esterification and aldol condensation are a few other examples of the dehydration reaction.
Mechanism of Dehydration Reaction
The removal of water from alcohol to form an alkene is an elimination reaction, more precisely E1 elimination. It is a two-step process involving the following [6]:
Step 1: Protonation of the -OH group to form -OH2+, a better leaving group than -OH. It is followed by the cleavage of the C-O bond, leaving a carbocation.
Step 2: Deprotonation of the carbocation by a water molecule, which acts as a base. It leads to the creation of a C=C bond. The resulting alkene is a stable product following Zaitsev’s rule.
Difference between Dehydration Synthesis and Hydrolysis
Hydrolysis is breaking large complex molecules into simpler ones by adding water along with an enzymatic catalyst. It is the opposite of dehydration synthesis and commonly occurs in the human body during food digestion. Polymers break down into monomers through hydrolysis. The bonds are broken, and energy is released. The results of dehydration synthesis can be reversed by hydrolysis [7].
Dehydration Synthesis vs. Hydrolysis
|
| Dehydration Synthesis | Hydrolysis |
|
Definition |
Bonds are formed through the removal of water |
Bonds are broken through the addition of water |
|
Mechanism |
Two molecules are joined covalently to form a longer molecule |
A molecule is split into smaller molecules |
|
Examples |
Formation of proteins from amino acids |
Breaking down of proteins into amino acids |
Dehydration Synthesis and Condensation Reaction
A condensation reaction is a type of chemical reaction in which two molecules or functional groups combine to form a larger molecule, accompanied by the loss of a small molecule. The small molecule can be water, hydrogen chloride, methanol, or acetic acid. Dehydration synthesis is a specific case of condensation reaction where two molecules condense to form a new product.
An example of a condensation reaction is polymerization, where a series of condensation steps result in a long-chained molecule. Intramolecular condensation is quite common in some molecules leading to a ringed structure.
FAQ
Ans. The reaction of bromelian and gelatin is hydrolysis.
Ans. Cellular respiration is neither dehydration synthesis nor hydrolysis.