Exothermic Reaction
Table of Contents
What is an Exothermic Reaction?
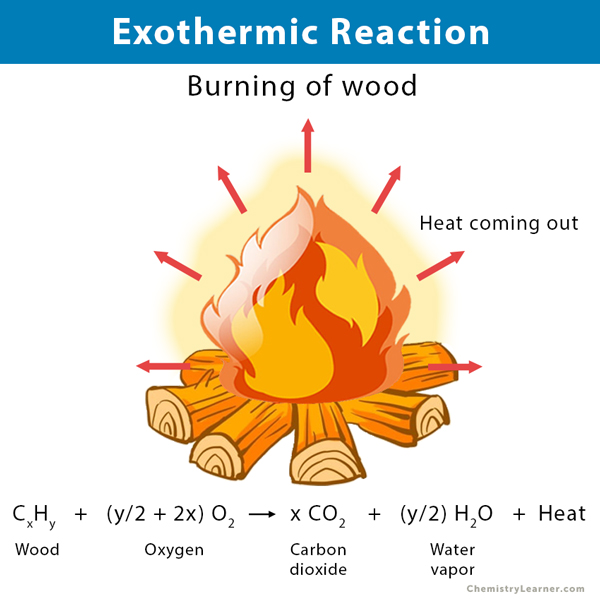
A chemical reaction is said to be exothermic when it releases energy in the form of heat. The system (reaction) releases heat to the surroundings as the reactants transform into products. For example, the burning of wood releases heat. An exothermic reaction causes the surroundings to heat up. That explains why we feel hot when we stand beside a fire. The exothermic reaction is the opposite of the endothermic reaction [1-3].
General Equation
Reactants → Products + Energy (heat)
Exothermic Process
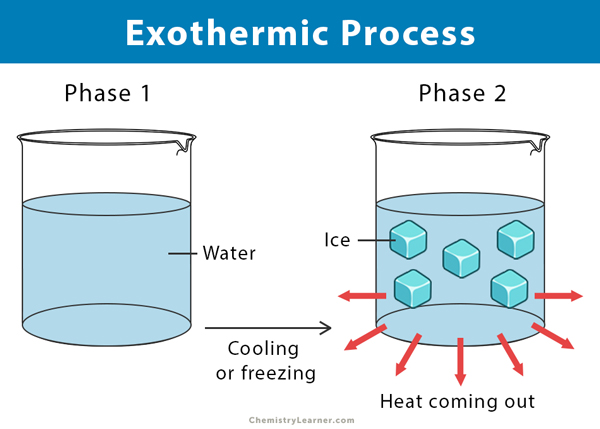
An exothermic process does not involve any chemical reaction. Here, a compound goes from one phase to another by releasing heat. For example, when water freezes into ice, heat is given out to the surroundings. Its temperature comes down until crystals of ice start to form.
Energy Diagram for Exothermic Reaction
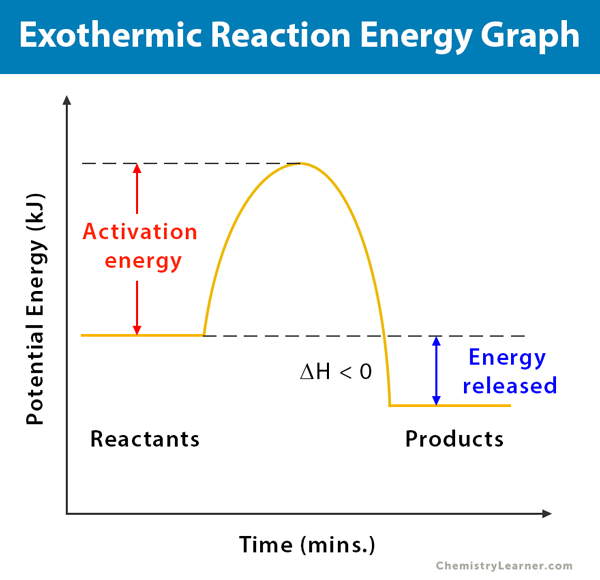
A chemical reaction always involves a change in energy. An energy diagram represents this change. An explanation of this graph is as follows. The potential energy of the reaction is plotted along the vertical axis, and the time of reaction is plotted along the horizontal axis. This graph shows the change in energy as the reaction progresses from reactants to products.
The enthalpy (H) of a reaction is a manifestation of energy. It represents the “heat content” of a reaction. Enthalpy change occurs when a reaction absorbs or emits heat at constant pressure. It is possible to calculate the enthalpy change (ΔH), which is defined as the difference between the products’ enthalpy and the reactants’ enthalpy. From the energy diagram of an exothermic chemical reaction, one can see that the reactants’ energy is higher than that of the products. Therefore, ΔH is negative (ΔH<0) and the reaction is exothermic. The energy diagram can tell if the reaction is exothermic or not.
During the reaction, few intermediates form. The activation energy of a reaction is the difference between the intermediates’ energy and reactants’ energy. In other words, the activation energy is the energy required for the reaction to overcome a “barrier” and proceed until the transformation is completed. Sometimes, a catalyst is used in a reaction to reduce the activation energy so that the reaction can proceed faster [4,5].
What Happens in an Exothermic Reaction?
In an exothermic reaction, the reactants are converted into products with the release of heat. Chemical bonds between the atoms break in the reactants, and new chemical bonds form in the products. The atoms rearrange to form the products.
The reactants have enough potential energy to overcome the energy barrier and transform into products. The energy generated by the reaction is given out to the surroundings.
An exothermic reaction is mostly spontaneous with a negative Gibbs’ free energy. However, its spontaneity depends on the temperature.
Examples and Applications of Exothermic Reaction and Process
Uses in Laboratory, Industry, and Power Plant
1. Exothermic Reaction
Here are some examples of exothermic reactions taking place in laboratories, industries, and power plants. The balanced chemical equations are shown along with the examples [1,5,6].
2 Na (s) + Cl2 (g) → 2 NaCl (s) + heat
- Calcium oxide (CaO) reacts vigorously with water (H2O) to produce calcium hydroxide (Ca(OH)2) or slacked lime.
CaO (s) + H2O (l) → Ca(OH)2 (aq.) + heat
- When calcium chloride (CaCl2) reacts with water (H2O), the products are hydrochloric acid (HCl) and calcium oxide (CaO).
CaCl2 (s) + H2O (l) → CaO (s) + 2 HCl (aq.) + heat
2 H2 (g) + O2 (g) → 2 H2O (l) + heat
- Thermite reaction: In this reaction, aluminum (Al) reduces iron (III) oxide (Fe2O3) to iron (Fe).
Al (s) + Fe2O3 (s) → Fe (s) + Al2O3 (s) + heat
- Synthesis of ammonia: Haber’s process is employed in the synthesis of ammonia. Here, nitrogen (N2) from the air combines with hydrogen (H2) derived mainly from natural gas (methane) to form ammonia (NH3).
N2 (g) + 3 H2 (g) → 2 NH3 (g) + heat
- Ammonia (NH3) dissolves in water (H2O) to form an ammonium cation (NH4+).
NH3 (g) + H2O (l) → NH4+ (aq.) + OH– (aq.) + heat
- Nuclear fission: When a neutron (n) collides with a heavy nucleus like uranium (235U), the collision releases a massive amount of energy (~200 MeV).
n + 235U → 139Ba + 96Kr + energy
- Nuclear fusion: Lighter elements combine to form new elements and release energy. Two deuterium (21H) atoms combine to produce helium-3 (32He).
21H + 21H → 32He + 10n + energy
- Sulfuric acid reacts with ordinary table sugar (sucrose) and results in an energetic exothermic reaction. The reaction produces heat, steam, carbon dioxide (CO2), and sulfur dioxide (SO2).
C12H22O11 (s) + H2SO4 (aq.) + ½ O2 (g) → 11 C (s) + 12 H2O (l) + CO2 (g) + SO2 (g) + heat
- Alkali metals, like sodium (Na), lithium (Li), and potassium (K), react rapidly with water (H2O), forming the corresponding hydroxide (OH) while releasing hydrogen (H2) gas.
2 Na (s) + 2 H2O (l) → 2 NaOH (aq.) + H2 (g) + heat
2. Exothermic Process
The following is an example of a process where no chemical reactions take place. The compound disassociates in water and releases heat.
- Dissolving sodium hydroxide (NaOH), also known as lye and caustic soda, in water (H2O) releases sodium ions (Na+) and hydroxide (OH–) ions along with heat.
NaOH (s) + H2O (l) → Na+ (aq.) + OH– (aq.) + H2O (l) + heat
Examples in Everyday Life
There are many useful examples of exothermic reactions and processes in everyday life.
- Snow formation: Snow is precipitation from the water vapor in the atmospheric clouds in the form of ice crystals. Snow falls from clouds as white flakes. When the temperature reaches below 0 ⁰C or 32 ⁰F, the water changes from liquid to solid (freezing).
H2O (l) → H2O (s) + heat
- Water vapor condensation: Water vapor present in clouds condenses when the temperature cools down and pours down like rain.
H2O (g) → H2O (l) + heat
- Deposition: When the temperature is below freezing, water vapor directly changes to ice without going through the water phase.
H2O (g) → H2O (s) + heat
- Respiration: Respiration is considered to be an exothermic reaction. Human beings consume carbohydrates and breathe oxygen. The glucose (C6H12O6) from carbohydrates reacts with the oxygen (O2) in the cells, releases carbon dioxide (CO2) and water (H2O), and provides energy to the human body.
C6H12O6 (s) + 6 O2 (g) → CO2 (g) + H2O (l) + energy
- Combustion reaction: Natural gas is commonly used in houses for cooking using a gas burner. The burning of any natural gas is a combustion reaction. When methane (CH4) is combusted in the presence of oxygen (O2), it gives heat, carbon dioxide (CO2), and water (H2O).
CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H2O (g) + heat
Burning a candle or matchstick is another example of a combustion reaction.
- Hot pack: A hot pack consists of water (H2O) and anhydrous calcium chloride (CaCl2) separated by a thin film and placed inside a plastic pack. When the pack is squeezed, the film breaks, and calcium chloride dissolves in water and releases heat.
CaCl2 (s) + H2O (l) → Ca+ (aq.) + 2 Cl– (aq.) + H2O (l) + heat
- Rusting of iron (Fe) in the presence of oxygen (O2) and moisture (H2O) to give iron (III) oxide (Fe2O3) is an oxidation reaction that releases heat.
4 Fe (s) + 3 O2 (g) + 2n H2O (g) → 2 Fe2O3.nH2O (s) + heat
- Adding water to laundry detergent releases heat that can be felt by placing the hands.
FAQ
Ans. An exothermic reaction can lead to thermal runaway when the heat produced exceeds the heat removed. As a result, the excess heat from the reaction is high enough to cause destructive effects.
Ans. All spontaneous processes are not exothermic. The reason is that Gibbs’ Free energy determines spontaneity and not enthalpy.
Ans. Neutralization reactions are generally exothermic. Bonds are being made, and energy is released to the surroundings.
Ans. No, not all decomposition reactions are exothermic since most decomposition requires energy to break bonds.