Maillard Reaction
What is the Maillard Reaction?
Maillard reaction is a chemical reaction that occurs between amino acids and reducing sugars. The thermal processing of amino acids and sugars produces a mixture of complex compounds. The reaction is responsible for the non-enzymatic browning of food, like a cookie, biscuit, bread, and steak. Browning occurs during frying, roasting, searing, and baking and affects the overall quality of food. It produces changes in sensory attributes such as color, flavor, and aroma. Aside, browning alters the nutritional properties and global recognition.
The reaction’s history goes back to 1912 when it was first reported by the French chemist Louis-Camille Maillard.
What Happens in Maillard Reaction?
For the Maillard reaction to occur, an amino (-NH2) group has to be present. An amino group is commonly found in proteins, peptides, and amino acids. Reducing sugar is a carbohydrate with a free aldehyde or ketone group capable of reducing other compounds. Simple sugars like monosaccharide (glucose and fructose) and disaccharides (lactose) go through the Maillard reaction. This reaction’s optimum temperature is 140 to 165 °C (284 to 329 °F). Many complex compounds that give distinct food flavor form during the reaction. Some of these compounds break down into simple compounds.
Acrylamide is a byproduct of the Maillard reaction. It is formed between asparagine, an α-amino acid found in food, and a dicarbonyl. Acrylamide is found in many foods. According to the International Agency for Research on Cancer (IARC), it is considered a carcinogen.
Factors Affecting Maillard Reaction
The Maillard reaction is affected by several factors, some of which are significant.
- Temperature: High temperatures, up to 180 °C (355 °F), increases the rate of reaction and result in water evaporation.
- Time: The browning of glucose and fructose during the reaction with the milk protein casein increases with heating time.
- pH: The loss of fructose in a fructose-lycine reaction increases with pH.
Aside, there are also a few minor factors, like:
- Water activity
- Reactant source
- Concentration
- Type and ratio of reducing sugar, amino acid, and food composition
Mechanism of Maillard Reaction
The mechanism of the Maillard reaction involves the following steps:
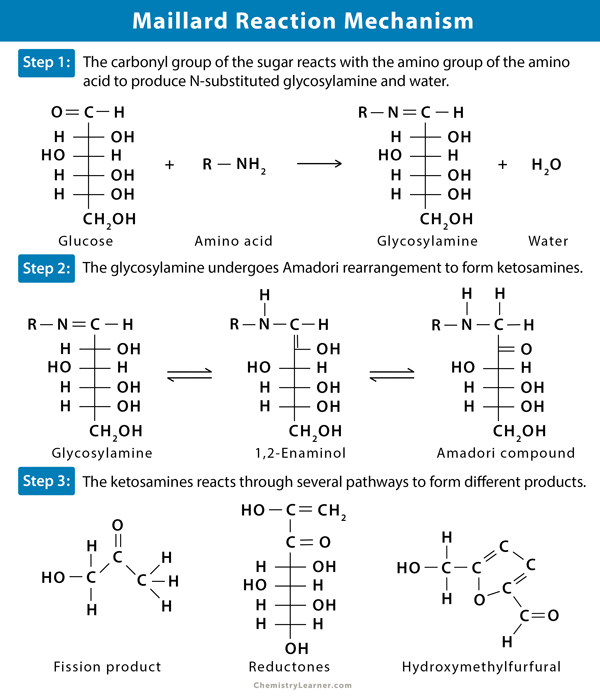
- The carbonyl group of the sugar reacts with the amino group of the amino acid. The result is N-substituted glycosylamine and water.
- The unstable glycosylamine undergoes Amadori rearrangement and forms ketosamines.
- The ketosamines further react through several pathways:
- Produce two molecules of water and reductones
- Form diacetyl, aspirin, pyruvaldehyde, and numerous fission products of the sugar–amino compound.
- Produce polymerized polymers and brown pigment, called melanoidins
Importance of Maillard Reaction
The Maillard reaction produces flavor and aroma during the cooking process. It is used everywhere, from the baking to the dairy industry. It has impacted our lives as the reaction makes food tasty. The roasted coffee flavor, the yellow-gold color of French fries, golden-brown color of bread, the umami taste in fried onions, and browning of steak when smeared or grilled are all results of the Maillard reaction.
Maillard Reaction vs. Caramelization
Caramelization also causes browning in food. However, the two are different processes of non-enzymatic browning. While the Maillard reaction involves amino acid, caramelization is merely the pyrolysis of sugar involving thermal decomposition. It gives food a sweet and nutty flavor. Examples of caramelization are the production of caramel candies, preparation of crème bûlée, and the making of caramelized onions, carrots, and apples.
The caramelization reaction temperature depends on the type of sugar. Sucrose and glucose caramelize around 160 °C (320 °F), and fructose caramelizes at 110 °C (230 °F).
FAQ
Ans. Pressure cookers create an ideal Maillard reaction since pressure cooking produces the browning effect without boiling the liquids. They make a deep, rich flavor infusion that is not possible in slow cooking.