Spontaneous and Non-Spontaneous Reaction
What is Spontaneous Reaction[1-5]
A spontaneous reaction is a chemical reaction that proceeds without any supply of energy. A reaction that is spontaneous will proceed without outside intervention. The Gibbs free energy of the products is lower than that of the reactants, which implies stable products. Therefore, the change in the free energy of the reaction is negative.
In chemistry and biology, a spontaneous reaction occurs naturally. It favors the formation of products under the given reaction conditions. An example of a spontaneous reaction is the exergonic reaction.
Spontaneous Process
In absence of a chemical reaction, a system can still undergo change. However, the change is only physical and the process is known as a spontaneous process. Examples of the spontaneous process include a ball rolling down a slide and ice melting into water.
Spontaneous Reaction and Gibbs Free Energy[1-4]
What Causes a Reaction to be Spontaneous
The spontaneity of a reaction occurring at constant temperature and pressure can be determined by Gibbs free energy, which is given by, [2-6]
ΔG = ΔH – TΔS
Here, ΔH is the change in enthalpy, ΔS is the change in entropy, and T is the temperature. ΔG is expressed in the unit of kJ mol-1. The sign of ΔG depends on the signs of ΔH and ΔS. A reaction is spontaneous and favorable if there is a decrease in enthalpy and an increase in entropy of the system.
In general, the conditions for a spontaneous reaction to occur are as follows:
1. When ΔG is negative, the reaction is spontaneous and proceeds in the forward direction.
2. When ΔG is positive, the reaction is not spontaneous in the forward direction but is spontaneous in the reverse direction.
3. When ΔG is zero, the reaction is in equilibrium. The concentrations of the reactants and products will remain the same.
Examples of Spontaneous Reaction[3,5,7]
1. Combustion reaction: When a fuel like ethanol (C2H5OH) combusts in presence of oxygen (O2), it gives off a large amount of heat and energy. The products of the combustion reaction are carbon dioxide gas (CO2) and water (H2O) at temperatures above 100°C. The reaction will progress in the forward direction until the fuel runs out.
C2H5OH (l) + 3 O2 (g) → 2 CO2(g) + 3 H2O (g)
2. Aeration: When a soda bottle is opened, carbonic acid (H2CO3) present in the soda decomposes to carbon dioxide (CO2) and water (H2O).
H2CO3 (aq.) → CO2 (g) + H2O (l)
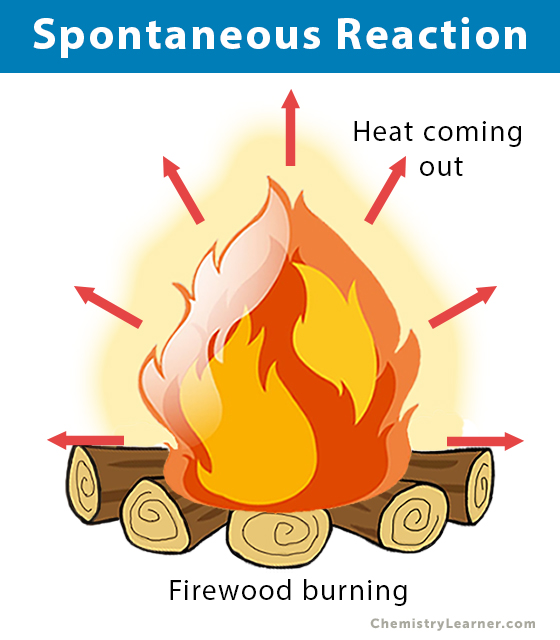
3. Burning of wood: Wood consists of hydrocarbon (CxHy). The burning of wood in the presence of oxygen (O2) produces carbon dioxide (CO2) and water (H2O).
CxHy + (x+y/4) O2 → x CO2 (g) + (y/2) H2O (l)
4. Baking soda and vinegar: The reaction of baking soda (NaHCO3) with vinegar (CH3COOH) gives sodium acetate (CH3COONa), carbon dioxide (CO2), and water (H2O).
NaHCO3 (s) + CH3COOH (aq.) → CH3COONa (aq.) + CO2 (g) + H2O (l)
5. Formation of rust: The natural rusting of iron (Fe) in the presence of oxygen (O2) gives iron (III) oxide (Fe2O3).
4 Fe (s) + 3 O2 (g) → 2 Fe2O3 (s)
What is Non-spontaneous Reaction [2,3]
A non-spontaneous reaction is a chemical reaction that does not take place on its own. It requires the influence of external factors in the form of energy or heat to proceed. When reactants are mixed, they do not transfer electrons to each other to form bonds. Energy must be supplied to drive the reaction forward. A non-spontaneous reaction is endothermic and is usually accompanied by an increase in Gibbs free energy.
Non-spontaneous Process
Suppose a chemical reaction does not take place in a system. In that case, the system can still take energy and undergo a physical process. Such a process is known as a non-spontaneous process. Melting ice into water and boiling water into steam are examples of non-spontaneous processes.
Examples of Non-spontaneous Reaction[3]
1. Electrolysis of water: Energy in the form of an electric current is supplied to water (H2O), which decomposes into hydrogen (H2) and oxygen (O2). This process is known as electrolysis and is a redox reaction.
2 H2O (l) → 2 H2 (g) + O2 (g)
Another example of non-spontaneous redox reactions is electroplating.
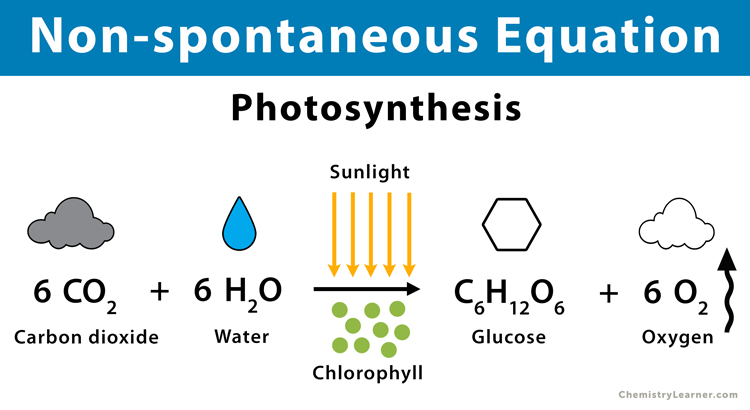
2. Photosynthesis: A photochemical reaction in which plants take in energy from the sun (hν) to convert carbon dioxide (CO2) and water (H2O) into sugar (C6H12O6) and oxygen (O2).
6 CO2 (g) + 6 H2O (l) + hν → C6H12O6 (s) + 6 O2 (g)
3. Ozone formation: Conversion of oxygen (O2) to ozone (O3) is a non-spontaneous process at all temperatures. It is driven by ultraviolet (hν) light from the sun.
3 O2 (g) + hν → 2 O3 (g)
Spontaneous vs. Non-spontaneous Reactions[2-4,6,12]
The following table gives the difference between spontaneous and non-spontaneous reactions.
| Spontaneous Reaction | Non-spontaneous Reaction | |
|---|---|---|
| Definition | A chemical reaction that takes place without any external energy | A chemical reaction that requires external energy to occur |
| Enthalpy and entropy change | Favors increasing entropy and decreasing enthalpy | Do not favor increasing entropy and decreasing enthalpy at normal conditions |
| Gibbs free energy | Negative | Positive |
| Type | Exergonic | Endergonic |
| Example | Combustion of wood and fuel | Electrolysis of water |
How to Tell if a Reaction is Spontaneous or Non-spontaneous[12]
The spontaneity of a reaction depends on the change in enthalpy (ΔH) and entropy (ΔS) as well as the temperature (T). The following table predicts whether a reaction will be spontaneous or not.
| ΔH < 0 | ΔH > 0 | |
| ΔS > 0 | Spontaneous at all T | Spontaneous at high T |
| ΔS < 0 | Spontaneous at low T | Non-spontaneous at all T |