Zeroth Law of Thermodynamics
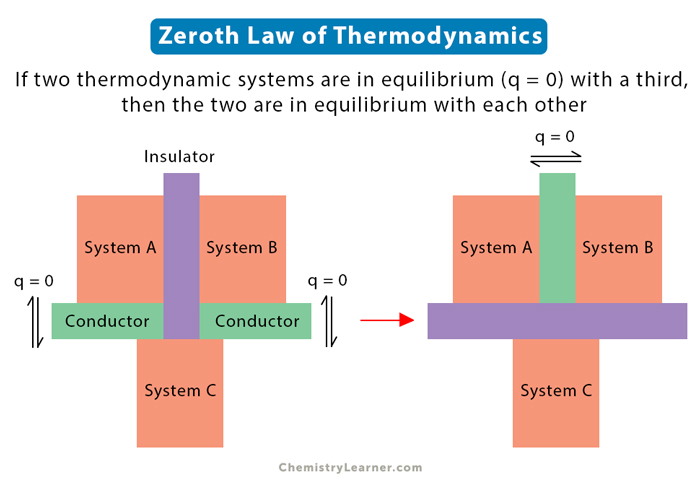
The zeroth law of thermodynamics states that if two systems are in thermal equilibrium with a third one, then the two are in thermal equilibrium with each other. It also means that the three systems are at the same temperature. The most remarkable part of the zeroth law is that it establishes temperature as an essential and quantifiable property of matter [1-4].
British physicist and astronomer Ralph Fowler formulated and labeled the zeroth law in 1931. This law was established after the first and second laws of thermodynamics. Because of its importance over the other two laws in understanding temperature, it was called the zeroth law.
Zeroth Law and Thermal Equilibrium
Consider two bodies at different temperatures that are in contact. Temperature decides whether heat exchange takes place between them or not. Heat will flow from the hotter body to the colder body. After some time, the heat stops flowing, and the two bodies are in thermal equilibrium. Thus, thermal equilibrium leads to the unabridged definition of temperature. When two objects are in thermal equilibrium, they are at the same temperature [1-6].
Examples and Applications [1-6]
1. The thermometer is the most common application of the zeroth law of thermodynamics. We can take an ordinary thermometer and observe the expansion of mercury inside as the temperature increases. We notice that the height of the mercury column increases, which indicates a temperature change. This height can be measured, and the exact temperature can be obtained.
2. When a glass of ice-cold water and a glass of boiling water is kept on the kitchen countertop, both reach equilibrium with the surrounding air, which is at room temperature. Thus, both glasses will be in thermal equilibrium at room temperature.
3. When a packet of frozen vegetables is kept in the freezer, it will reach thermal equilibrium with the freezer and other items inside the freezer. The packet and the other items will be at the freezer’s temperature.
FAQs
Q.1. What are the limitations of the zeroth law of thermodynamics?
Ans. The limitation of the zeroth law of thermodynamics is that it cannot be derived from other thermodynamics laws. It does not tell us how the heat is transferred, or energy is converted.