Molecular Geometry
Molecular geometry refers to the arrangement of atoms in a molecule in a two- or three-dimensional structure. A molecule consists of a central atom chemically bonded to several side atoms, resulting in a well-defined shape and a finite bond angle [1-4].
Molecular geometry is vital in chemistry to determine a substance’s physical and chemical properties, like color, reactivity, polarity, and state of matter.
VSEPR Theory and Molecular Geometry
Molecular geometry is generally studied using the VSEPR theory, an abbreviation for valence shell electron pair repulsion. It predicts a molecule’s shape based on the number of bonds and lone pairs. According to this theory, the lone pairs in the valence shell of the central atom will rearrange themselves in such a way as to minimize the repulsion and maximize the distance between them. As a result, the molecule forms a regular geometric shape. The angle between the adjacent bonds defines the bond angle [1-11].
Notation
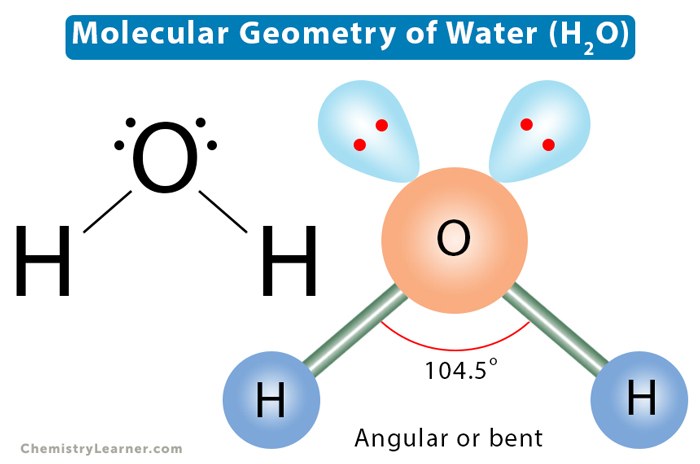
According to VSEPR theory, a molecule is designated by the letters AXm. “A” represents the central atom. “X” represents the surrounding atoms, and “m” is the number of electron groups associated with the central atom. An electron group consists of a single, double, or triple bond. If lone pairs are present, the letter En is added, where “n” represents the number of lone pairs surrounding the central atom. For example, the water (H2O) molecule has two bonded and two lone pairs. Its VSEPR notation is AX2E2.
Shape of Molecules
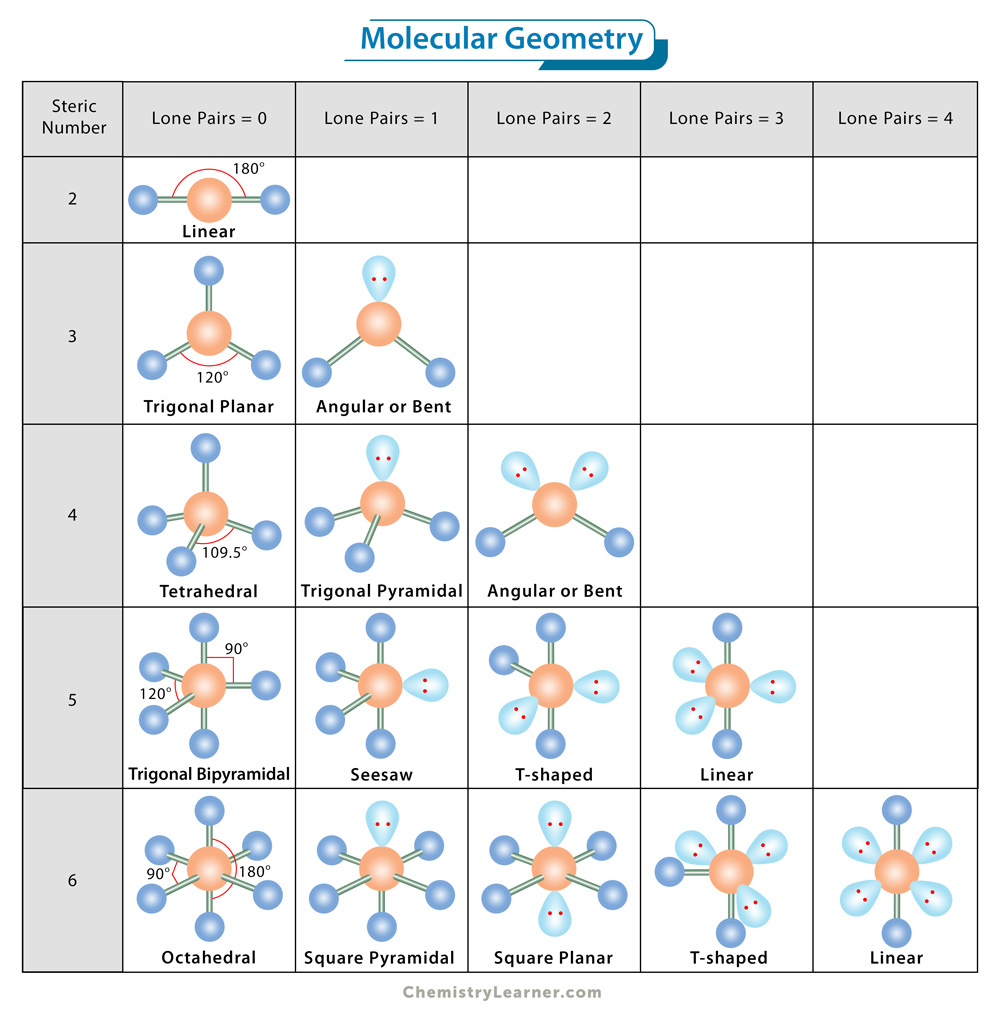
First, we shall consider the molecular shape without lone pairs on the central atom. The VSEPR theory describes five main shapes of simple molecules. They are as follows [1-11]:
1. Linear: It specifies the geometry shaped by a central atom surrounded by two other atoms. The atoms are arranged in a straight line, and the angle between the bonds, or bond angle, is 180 °. The VSEPR notation is AX2. Examples of molecules with linear geometry are carbon dioxide (CO2), beryllium chloride (BeCl2), and nitric oxide (NO).
2. Trigonal Planar: The molecule forms a triangular shape in one plane. It has one atom at the center and three at the corners of an equilateral triangle, making a bond angle of 120°. The VSEPR notation is AX3. Examples are boron trifluoride (BF3), boron trichloride (BCl3), and sulfur trioxide (SO3).
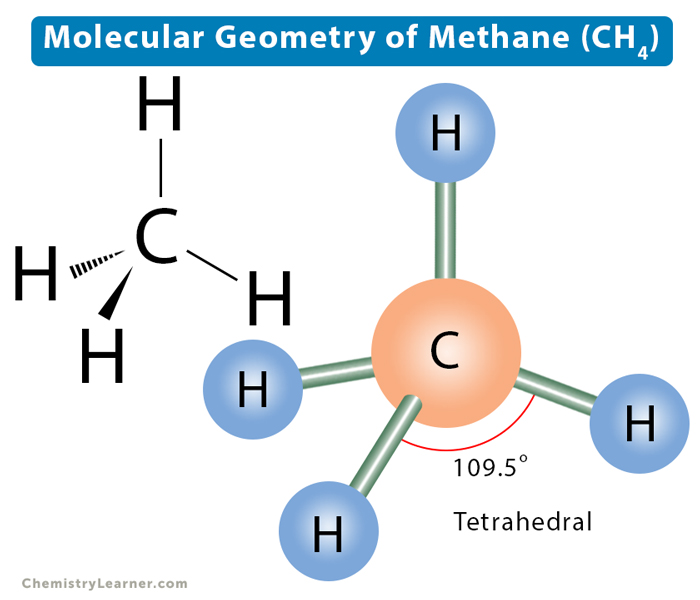
3. Tetrahedral: This shape occurs when one atom occupies the center, and four others are located at the corners of a tetrahedron. This shape is because the four bond pairs experience minimum repulsion when the bonds are directed toward the corners of the tetrahedron. The angle between the bonds is 109.5°. The VSEPR notation is AX4. An example is methane (CH4).
4. Trigonal Bipyramidal: The molecule’s shape resembles a pyramid with a triangular base. Unlike the trigonal planar, the trigonal bipyramidal structure is three-dimensional. The central atom is surrounded by five atoms – three in one plane and two on opposite sides of the plane. The bond angle is 90° in one plane and 120° in another. The VSEPR notation is AX5. Examples are phosphorous pentachloride (PCl5) and arsenic pentafluoride (AsF5).
5. Octahedral or square bipyramidal: It describes the shape of compounds with six atoms or groups of atoms or ligands symmetrically arranged around a central atom such that they are positioned at the vertices of an octahedron. The octahedral shape is characterized by bond angle values of 90° and 180°. The VSEPR notation is AX6. Examples are sulfur hexafluoride (SF6) and molybdenum hexacarbonyl Mo(CO)6.
Lone Pairs in Molecular Geometry
Nonbonding electrons or lone pairs on the central atom affect the molecular shape. The lone pairs are located in the atomic orbitals of the central atom and repel other orbitals, causing a deviation from the abovementioned geometry [1-11].
Lone pairs are assumed to have a more significant repulsive effect than bonding pairs. They are closer to the central atom’s nucleus and spread over a larger space than bonding electrons. Therefore, they tend to have the least amount of interaction with the bonding pairs.
The negatively-charged space around the lone pairs behaves similarly to that associated with the bonded atoms. The valence shell orbitals containing the bonding and nonbonding pairs will spread out from the central atom in directions that minimize their mutual repulsions. The repulsion behavior among the lone and bond pairs follows the following pattern.
lone pair-lone pair > lone pair-bond pair > bond pair-bond pair
Examples
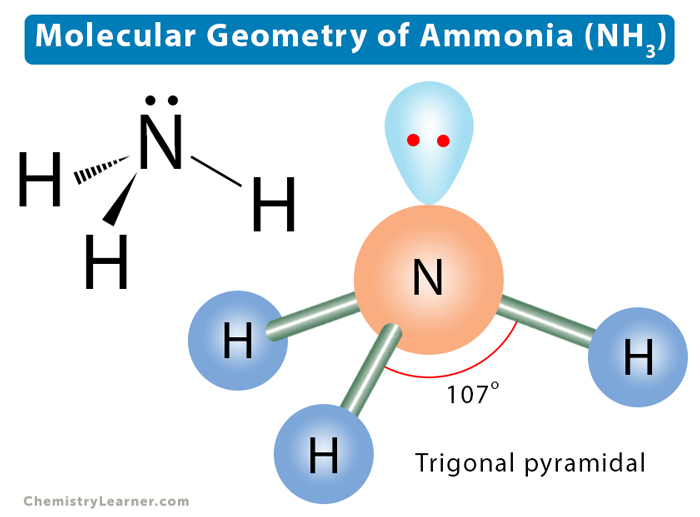
In the discussion above, we found four electron pairs distributed in a tetrahedral shape. Lone pairs on the central atom can explain the changes in molecular shape and bond angle as we go from methane to ammonia to water [5].
Methane (CH4) consists of all bond pairs. Therefore, its shape is tetrahedral with a standard bond angle of 109.5°.
Ammonia (NH3) consists of three bond pairs and one lone pair. The lone pair will push the bond pairs outwards, reducing the bond angle to 107°. The resultant molecular shape is trigonal pyramidal.
Water (H2O) has two bond pairs and two lone pairs. The two lone pairs will repel the bond pairs further, thereby reducing the bond angle to 104.5°. The overall shape of the molecule is bent or angular.
Other examples include
Molecular Geometry Chart
The following table lists all the molecular geometries, the number of bond and lone pairs, and examples of each geometry type [5].
| Number of Electron Pairs from Lewis Structure | Number of Electron Groups | Number of Lone Pairs | Molecular Geometry | Examples |
|---|---|---|---|---|
| 2 | 2 | 0 | Linear | BeCl2, CO2, N3– |
| 3 | 3 | 0 | Trigonal planar | BCl3, SO3, CO32– |
| 3 | 2 | 1 | Angular or bent | SO2, O3, NO2– |
| 4 | 4 | 0 | Tetrahedral | CH4, NH4+, PO43– |
| 4 | 3 | 1 | Trigonal pyramidal | H3O+, NH3, XeO3 |
| 4 | 2 | 2 | Angular or bent | H2O, NH2–, ClO2– |
| 5 | 5 | 0 | Trigonal bipyramidal | PCl5, PF5 |
| 5 | 4 | 1 | “See-saw” | SF4, PBr4– |
| 5 | 3 | 2 | T-shaped | ClF3, XeF3+ |
| 5 | 2 | 3 | Linear | ICl2–, XeF2 |
| 6 | 6 | 0 | Octahedral | SF6, Mo(CO)6 |
| 6 | 5 | 1 | Square pyramidal | IF5, SF5–, SbF52– |
| 6 | 4 | 2 | Square planar | ICl4–, XeF4 |