Adsorption
Table of Contents
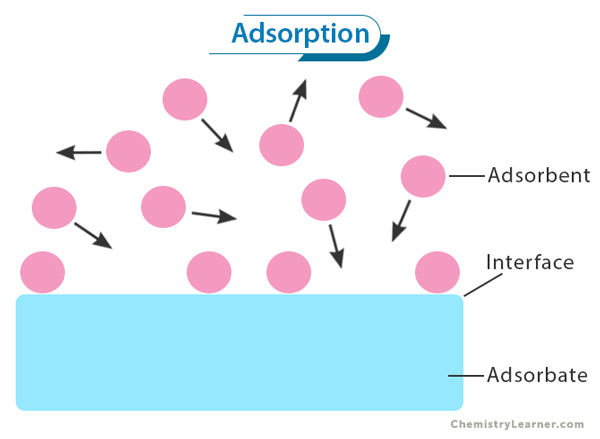
Adsorption refers to the process of adhesion of molecules or ions to a solid surface. It is the phenomenon where substances from a fluid phase, such as gases or liquids, accumulate on the surface of a solid material. [1-4]
Adsorption is driven by attractive forces between the adsorbate (the substance being adsorbed) and the adsorbent (the solid material). These attractive forces can be physical or chemical, depending on the specific system. Intermolecular forces include van der Waals forces, electrostatic interactions, and hydrogen bonding.
Adsorption finds diverse applications across several industries due to its ability to remove impurities, purify substances, and facilitate various processes.
Types of Adsorption
There are two types of adsorption – physical adsorption and chemical adsorption. [1]
Physical Adsorption
In physical adsorption or physisorption, weak van der Waals forces are responsible for attracting and holding the molecules or atoms onto the surface of the adsorbent. These intermolecular forces are relatively weak compared to chemical bonds. The physical adsorption process is reversible and depends on temperature and pressure.
Example
One example of physical adsorption is the adsorption of gases, like CO2, H2, and N2, on activated charcoal. Adsorption on activated charcoal is extensively used in air purification systems, gas masks, and even medical applications to adsorb toxins and odors from the air.
Chemical Adsorption
Unlike physisorption, which is characterized by weak van der Waals forces, chemical adsorption or chemisorption results in chemical bonds. The adsorbate species undergoes a chemical reaction with the adsorbent surface. This process requires a certain amount of energy to overcome the activation energy barrier. The rate at which chemisorption occurs is governed by rate constants, which depend on temperature and pressure.
Example
A classic example of chemical adsorption is the adsorption of gases on solid surfaces, such as hydrogen gas adsorption on a metal catalyst like platinum or palladium. This adsorption process is crucial in various industrial applications, especially in catalytic converters used in automobiles, where these metals facilitate the conversion of harmful gases like carbon monoxide and nitrogen oxides into less toxic substances through chemical reactions.
Adsorption Isotherm
Adsorption isotherm describes the relationship between the amount of adsorbate molecules adsorbed onto a solid surface and the equilibrium pressure or concentration of the adsorbate in the surrounding medium. [1,3]
A commonly used adsorption isotherm is the Langmuir Isotherm, which was proposed by Irving Langmuir in 1918. The Langmuir model assumes that adsorption occurs on a homogeneous surface with a limited number of identical sites. It suggests that once an adsorbate molecule occupies a site on the surface, no further molecules can be adsorbed at that site. It leads to the formation of a monolayer coverage.
Another widely applied isotherm is known as Freundlich Isotherm, proposed by Herbert Freundlich in 1909. Unlike Langmuir’s assumption of monolayer coverage, Freundlich’s model allows for multilayer coverage and heterogeneous surfaces. It describes non-ideal behavior by introducing an empirical constant related to surface heterogeneity.
The Brunauer-Emmett-Teller (BET) theory provides another perspective on adsorption isotherms. It was developed by Stephen Brunauer, Paul Hugh Emmett, and Edward Teller in 1938 to describe gas physisorption on solid surfaces with multilayer coverage. The BET theory assumes that each layer beyond the first exhibits identical properties and follows Langmuir-like behavior.
Adsorption vs. Absorption
The primary difference between adsorption and absorption is that adsorption occurs when particles adhere to the surface of a substance, while absorption involves the transfer of particles into another material (as one substance absorbs another). Here is a table highlighting the key differences between the two. [3-5]
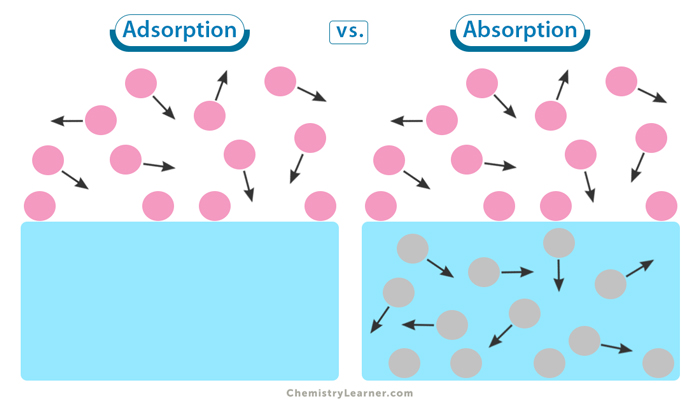
| Aspect | Adsorption | Absorption |
|---|---|---|
| Definition | Adhesion of particles onto a substance’s surface | Transfer of particles into another material |
| Process | Occurs at the surface of a substance | Transfer of particles into another material |
| Depth of Action | Surface level | Throughout the entire material |
| Reversibility | Usually reversible | Often irreversible |
| Energy Release | Often releases less energy | Can release significant energy during the process |
| Examples | Activated carbon, catalysts | A sponge soaking up water, salt dissolving in water |