Aldol Reaction
Definition: What is Aldol Reaction?
Aldol reaction is an essential class of chemical reaction, occasionally encountered in biochemistry. In an aldol reaction, new carbon-carbon bonds are forms due to the reaction between two carbonyl compounds. The product formed is a β-hydroxy carbonyl compound, also called an aldol product (aldehyde + alcohol). The aldol reaction is used to join two identical aldehydes or ketones together (dimer), or two different aldehydes or ketones (crossed-aldol). In order to avoid the production of multiple products, the two different aldehydes or ketones must be chosen carefully. High yields of crossed-aldol products can be synthetically useful if one uses an enolizable ketone, i.e., having α-hydrogens, and a non-enolizable aldehyde, i.e., without α-hydrogen. The enol is the nucleophile in the aldol reaction because it has pi electrons, and the electrophilic aldehyde is reactive enough to avoid the self aldol reaction of the ketone. When the nucleophile and electrophile are different, the reaction is called a crossed or mixed aldol reaction. On the contrary, when the nucleophile and electrophile are the same, the reaction is called an aldol dimerization [1-5].
These aldol products can often then undergo dehydration (loss of water) to give conjugated systems (elimination reaction). This reaction is then called an aldol condensation or Claisen-Schmidt condensation.
The history of the aldol reaction goes back to 1872 when French chemist Charles Wurtz first prepared the β-hydroxy aldehyde from acetaldehyde.
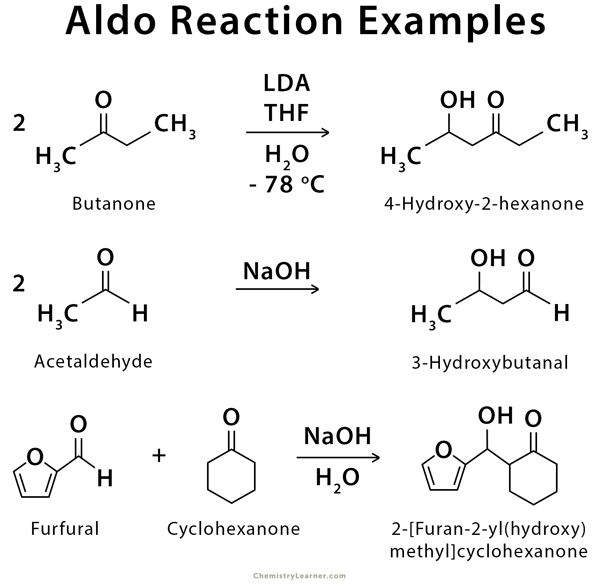
Examples of Aldol Reaction
Examples of aldol reaction include the addition reaction of aldehydes and ketones to form dimers and mixed or crossed-aldol products [1-3,6-8].
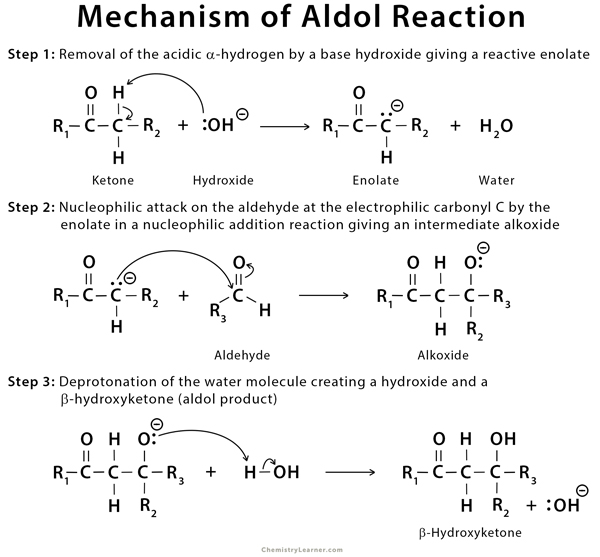
Mechanism of Aldol Reaction
The mechanism of aldol reaction involves the formation of carbanion (enolate), which attacks the carbonyl carbon of another molecule of a carbonyl compound to form intermediate, which is protonated further to form the product (aldol) [4,6,7,9].
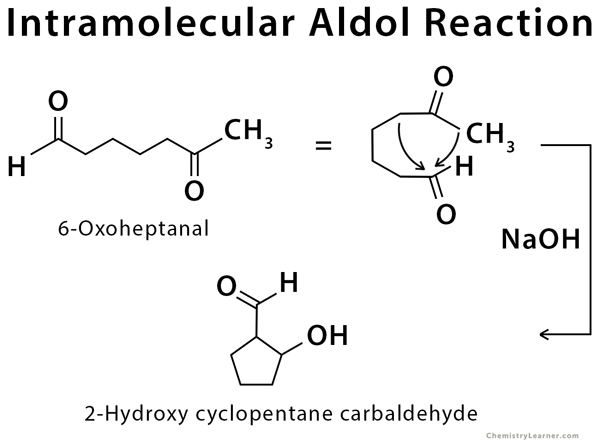
Intramolecular Aldol Reaction
Intramolecular aldol reaction happens when a single molecule contains two reactive aldehyde or ketone groups (dicarbonyl compound). The α-carbon of one group attacks the other, and the molecule attacks itself, forming a five- or six-membered ring structure.
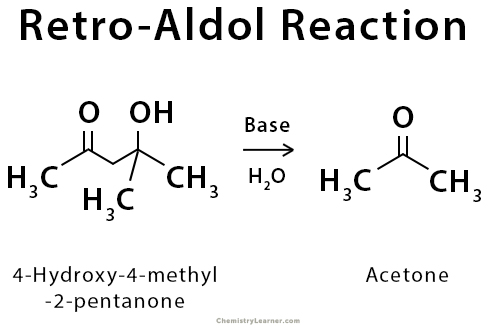
Retro-Aldol Cleavage Reaction
The retro-aldol cleavage reaction is the reverse of aldol reaction. In this reaction, a β-hydroxy carbonyl compound decomposes into an aldehyde or ketone and another carbonyl compound.
Uses and Applications of Aldol reaction
Structural aldol products are found in many essential molecules, whether naturally occurring or synthetic. For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritol and the synthesis of the heart disease drug Lipitor.
Difference between Aldol Reaction and Aldol Condensation
The aldol reaction is the first step in the aldol condensation. In the aldol reaction, an enolate nucleophile adds to an aldehyde or ketone to form a β-hydroxy carbonyl compound, called an aldol. In the aldol condensation, that compound can then undergo elimination to form an α,β-unsaturated carbonyl compound.
FAQs
Ans. Aldol reaction is the addition of enolates to aldehydes or ketones, and Claisen condensation is the addition of enolates to esters. It follows the same first two steps as that of the Aldol reaction.
Ans. The Mukaiyama aldol addition is an organic reaction and a type of aldol reaction between a silyl enol ether and an aldehyde or formate.
References
- Definition and example – Chemistry.msu.edu
- Definition and example – Chem.ucla.edu
- Definition and example – Chem.libretexts.org
- Definition and mechanism – Chem.libretexts.org
- Definition – Leah4sci.com
- Example and mechanism – Chem.libretexts.org
- Example and mechanism – Chem.ucalgary.ca
- Example – Butane.chem.uiuc.edu
- Mechanism – Chemistry2.csudh.edu