Arrhenius Theory
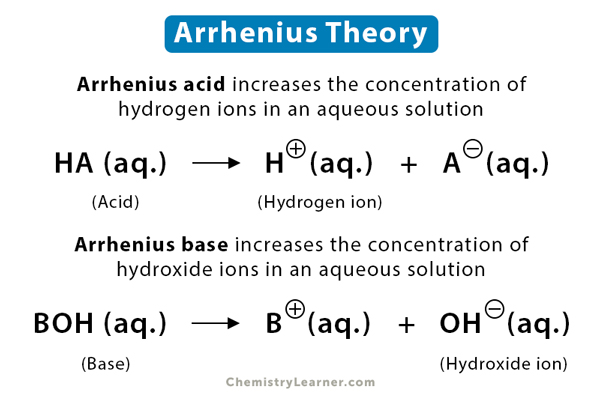
Arrhenius theory defines acids and bases in aqueous solutions. Swedish scientist Svante Arrhenius introduced the theory in 1887. According to this theory, an acid is a substance that gives off hydrogen ions (H+) in an aqueous solution, and a base produces hydroxide ions (OH–) in an aqueous solution. The hydrogen ion cannot exist alone in a water solution. Instead, it combines with a water molecule to form the hydronium ion (H3O+) [1-4].
The behavior of many well-known acids, such as sulfuric acid, nitric acid, and acetic acid, and many well-known bases, like sodium hydroxide, potassium hydroxide, and calcium hydroxide, can be explained by Arrhenius theory. Further, such acids and bases can be classified as strong or weak, depending on the hydrogen ion or hydroxide ion concentrations observed in solutions. The reaction between an acid and a base will produce salt and water. The latter is produced from the reaction between hydrogen ion and hydroxide ion.
Examples
Arrhenius Acid
The dissociation of hydrochloric acid (HCl) results in a hydrogen ion (H+) and a chloride ion (Cl–).
HCl (aq.) → H+ (aq.) + Cl– (aq.)
Since this reaction increases H+ ion concentration in the solution, hydrochloric acid is an Arrhenius acid.
Note:
In aqueous solutions, there are no free H+ ions. They react with water to form hydronium ions (H3O+).
H+ (aq.) + H2O (l) → H3O+ (aq.)
Therefore, the dissociation of HCl can be written more accurately as
HCl (aq.) + H2O (l) → H3O+ (aq.) + Cl– (aq.)
Other examples of Arrhenius acids are as follows:
1. Hydrofluoric acid (HF) dissociates into a hydronium ion and a fluoride ion (F–).
HF (aq.) + H2O (l) → H3O+ (aq.) + F– (aq.)
2. Hydrobromic acid (HBr) dissociates into a hydronium ion and a bromide ion (Br–).
HBr (aq.) + H2O (l) → H3O+ (aq.) + Br– (aq.)
3. Nitric acid (HNO3) dissociates into a hydronium ion and a nitrate ion (NO3–).
HNO3 (aq.) + H2O (l) → H3O+ (aq.) + NO3– (aq.)
4. Sulfuric acid (H2SO4) dissociates into two hydronium ions and a sulfate ion (SO42-).
H2SO4 (aq.) + 2 H2O (aq.) → 2 H3O+ (aq.) + SO42- (aq.)
Arrhenius Base
Highly soluble sodium hydroxide (NaOH) dissociates in water into a sodium ion (Na+) and a hydroxide ion (OH–) as follows:
NaOH (aq.) → Na+ (aq.) + OH– (aq.)
The presence of excess OH– ions makes it an Arrhenius base.
Other examples of Arrhenius base include:
1. Lithium hydroxide (LiOH) dissociates into a lithium ion (Li+) and a hydroxide ion.
LiOH (aq.) → Li+ (aq.) + OH– (aq.)
2. Barium hydroxide (Ba(OH)2) dissociates into a barium ion (Ba2+) and two hydroxide ion.
Ba(OH)2 (aq.) → Ba2+ (aq.) + 2 OH– (aq.)
3. Calcium hydroxide (Ca(OH)2) dissociates into a calcium ion (Ca2+) and two hydroxide ions.
Ca(OH)2 (aq.) → Ca2+ (aq.) + 2 OH– (aq.)
4. Ammonium hydroxide (NH4OH) dissociates into an ammonium ion (NH4+) and a hydroxide ion.
NH4OH (aq.) → NH4+ (aq.) + OH– (aq.)
Reaction Between Arrhenius Acid and Base
When Arrhenius acid reacts with Arrhenius base, we get salt and water. This reaction is known as an acid-base reaction or neutralization reaction.
Example
The reaction between hydrogen fluoride (HF) and lithium hydroxide (LiOH) results in lithium fluoride (LiF) and water (H2O). Let us look at the reaction step-by-step.
HF dissolves in water to form a hydrogen ion (H+) and a fluoride ion (F–).
HF (aq.) → H+ (aq.) + F– (aq.)
LiOH dissolves in water to form a lithium ion (Li+) and a hydroxide ion (OH–).
LiOH (aq.) → Li+ (aq.) + OH– (aq.)
Li+ and F– combine to form LiF, which is a salt. H+ and OH– react to form water. The overall reaction is given by
HF (aq.) + LiOH (aq.) → LiF (aq.) + H2O (l)
Limitations
1. The Arrhenius theory only describes acids and bases in their aqueous solutions. However, the acid-base reaction also occurs in gaseous phases. Arrhenius theory fails to explain such reactions. To overcome this limitation, many scientists prefer to use the Brønsted-Lowry theory.
2. According to Arrhenius theory, the presence of H+ ion and OH– ion is essential in the chemical reaction. However, a compound like aqueous sodium bisulfate (NaHSO4) is acidic, and aqueous sodium carbonate (Na2CO3) is basic. The former does not have an H+ ion, and the latter does not have an OH– ion.
3. The reaction between aqueous ammonia (NH3) and hydrochloric acid (HCl) yields ammonium chloride (NH4Cl) salt.
NH3 (aq.) + HCl (aq.) → NH4Cl (aq.)
However, there is no hydroxide ion in the reaction. Hydroxide ion is produced from the reaction between ammonia and water in an aqueous ammonia solution.
NH3 (aq.) + H2O (l) ⇋ NH4+ (aq.) + OH– (aq.)
The yield of hydroxide in the above reversible reaction is very low. Whatever hydroxide is present, it reacts with hydrogen ions to form water, which satisfies Arrhenius theory. However, a significant part of the reaction is between ammonia and hydrogen ions, which does not satisfy Arrhenius theory.