Avogadro’s Law
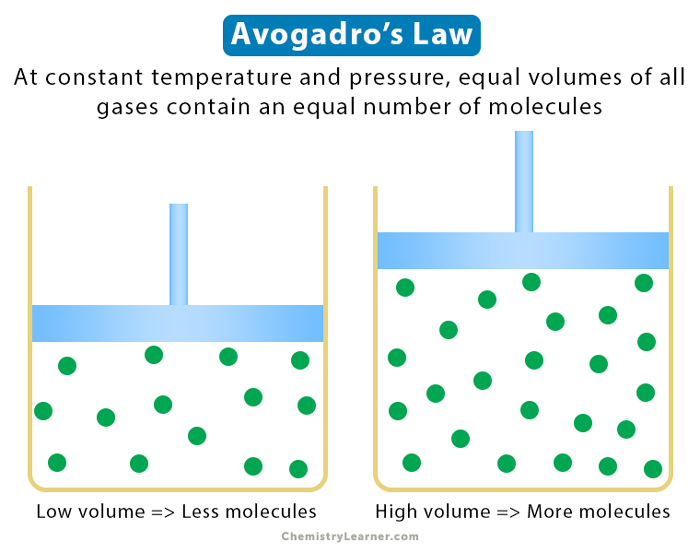
Avogadro’s law states that equal volumes of different gases contain an equal number of molecules under the same pressure and temperature conditions. This law is valid for ideal gases at low pressures and high temperatures [1-4].
Italian physicist Amedeo Avogadro was the first to state the hypothetical law in 1811.
Avogadro’s Law Formula
According to Avogadro’s hypothesis, the volume (V) of a gas is directly proportional to the number of moles (n) [1-4].
V ∝ n
Where
V : Volume
n : Number of moles
The above proportionality can be written as follows:
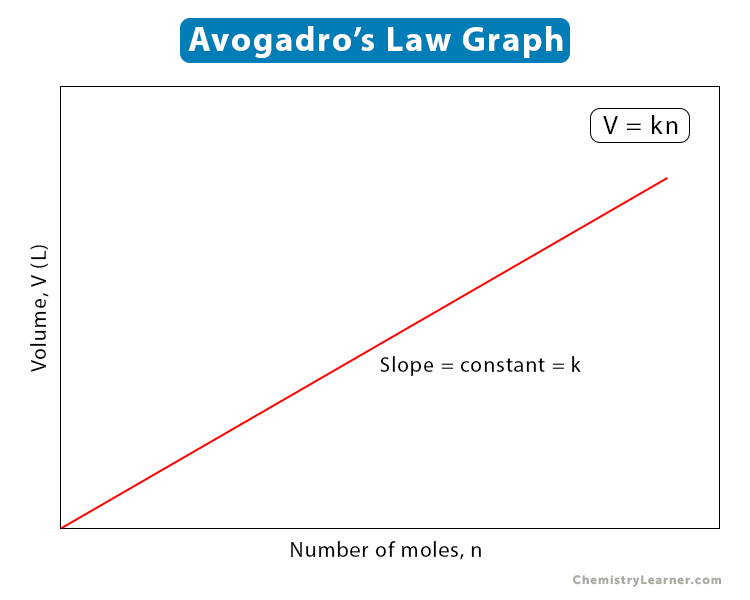
V = kn
Where
k : proportionality constant
This equation can be represented as a graph.
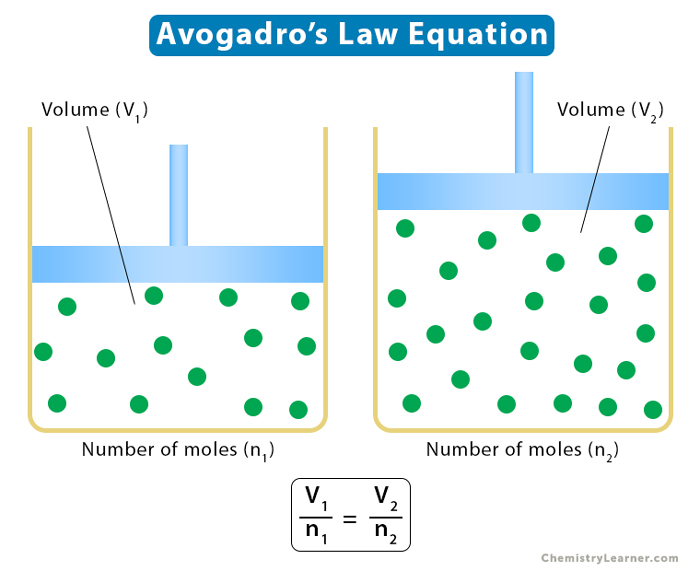
Consider a gas with n1 moles with a volume V1. Suppose the number of moles increases to n2 such that the volume increases to V2, then
V1 = kn1 and V2 = kn2
Dividing one equation by the other and rearranging,
V1/n1 = V2/n2
The above equation can be used to compare two gases at the same temperature and pressure conditions. Also, the number of molecules in a given volume of an ideal gas is independent of their size or the molar mass.
Derivation
In order to derive Avogadro’s gas law, let us look at the ideal gas equation.
PV = nRT
Or, V/n = RT/P
Where
P : Pressure
V : Volume
n : Number of moles
R : Universal gas constant
T : Temperature
At constant pressure and temperature, the right-hand side is constant. Let, k = RT/P. Then,
V/n = k
Thus, the volume is proportional to the number of moles of the gas.
Avogadro’s Number
Let us rewrite the ideal gas law as follows:
PV = (N/NA)RT
The ratio N/NA gives the number of moles or n = N/NA. Here, N is the number of molecules in the gas, and NA is known as Avogadro’s number. It is the number of molecules present in one mole of a substance. Its value is 6.023 x 1023.
Molar Volume
Avogadro’s number can be used to calculate the molar volume. For one mole of a gas, n = 1 and V = k. Therefore, one has to know the value of k at standard temperature and pressure (STP). At STP, the pressure (P) is 101.325 kPa, and the temperature (T) is 273.15 K. The universal gas constant R has a value of 8.314 L·kPa·M-1·K-1. Putting in all the values, we get
k = RT/P
Or, k = 8.314 L·kPa·M-1·K-1 x 273.15 K /101.325 kPa
Or, k = 22.4 LM-1
Examples
Below are some examples of Avogadro’s law in everyday life [5].
- Respiration: When we respire, we breathe in oxygen. The more we breathe, the more our lungs will expand.
- Deflation: When the air is released from an inflated tire, the number of moles decreases. The shape of the tire also changes since its volume decreases.
- Inflation: When a water tube is inflated by pumping air inside, the number of air molecules increases. Thus, the tube is inflated, and its volume increases.
Applications and Uses [1]
- Explains Gay-Lussac’s law
- Determines the atomicity of the gases
- Determines the molecular formula of a gaseous compound
- Gives the relationship between gram molecular mass and gram molecular volume of gas
- Determines the relationship between molecular mass and vapor density of a gas
Problems and Solutions
Problem 1: Two moles of helium gas fill up an empty balloon to a volume of 2.5 L. What would be the volume of the balloon if an additional 1.5 moles of helium gas is added at constant temperature and pressure.
Solution:
Given
n1 = 2 mol
V1 = 2.5 L
n2 = 2 mol + 1.5 mol = 3.5 mol
From Avogadro’s law,
V1/n1 = V2/n2
Therefore, the final volume of the balloon is
V2 = V1·n2 /n1 = (2.5 L x 3.5 mol)/2 mol = 4.375 L
Problem 2: 40 g of nitrogen gas is kept in a 2.5 L container. The gas exerts a pressure of 2 atm on the container. If pressure is kept constant, what is the final amount of gas in grams present in the container if gas is added until the volume has increased to 4.0 L?
Solution:
Given
Mass of nitrogen = 40 g => n1 = Mass/Molar mass = 40 g/28 gM-1 = 1.43 M
V1 = 2.5 L
V2 = 4 L
From Avogadro’s law,
V1/n1 = V2/n2
Or, n2 = n1 x V2/V1
Or, n2 = 1.43 M x 4 L/2.5 L
Or, n2 = 2.29 M
Therefore, amount of nitrogen
= 2.29 M x 28 gM-1 = 64 g





Dat nice
????????
this is the best explanation of Avogadro’s law. I love this!!!!!!!