Baeyer-Villiger Oxidation
Definition: What is Baeyer-Villiger Oxidation?
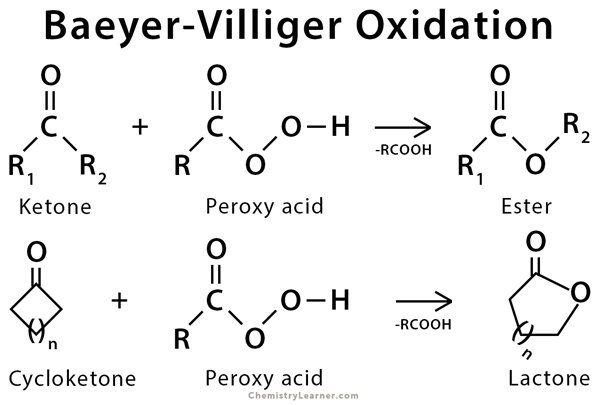
The Baeyer-Villiger oxidation is the oxidative cleavage of a carbon-carbon bond that is adjacent to a carbonyl functional group. This reaction is used to convert ketones to esters and cyclic ketones to lactones. It can be carried out with peroxy acids (also called peracid) like MCPBA, or with hydrogen peroxide and a Lewis acid. This reaction can be viewed as the insertion of O into one of the C-C bonds adjacent to the carbonyl functional group. For non-symmetrical cyclic ketones, the more highly substituted alkyl group migrates and becomes attached to the inserted O atom [1-5].
The history of this reaction goes back to 1899 when German chemists Adolf von Baeyer and Victor Villiger reported it.
Examples of Baeyer-Villiger Oxidation
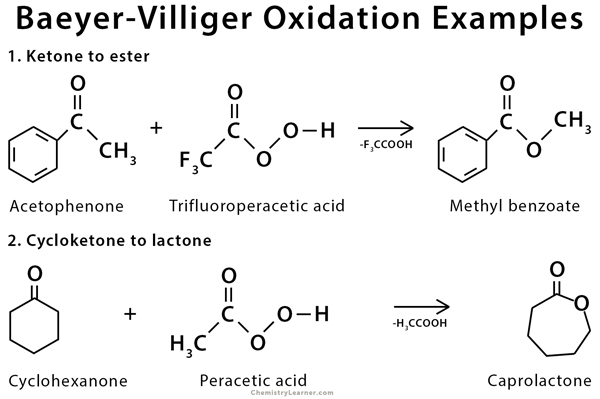
Baeyer-Villiger oxidation is used in the synthesis of caprolactone from cyclohexanone [2,6].
Mechanism of Baeyer-Villiger Oxidation
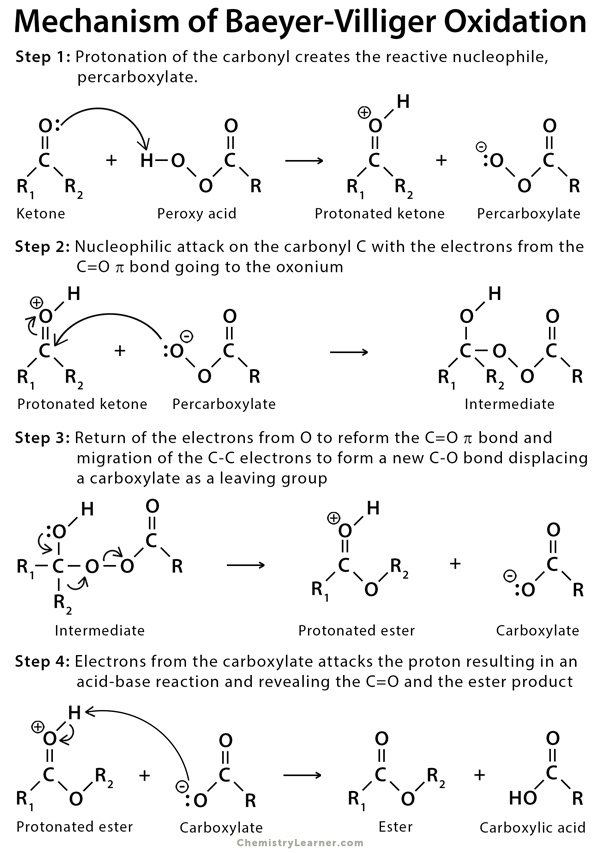
In this reaction, the ketone is oxidized, whereas the peroxy acid is reduced [6].
Application of Baeyer-Villiger Oxidation
Baeyer-Villiger oxidation is used in the synthesis of lactones, which contribute significantly to the flavor of fruits, and unfermented and fermented dairy products. Hence, they are used for flavor and fragrance. Polycaprolactone is a biodegradable polyester from which polyurethane is made.
References
- Definition – Organic-chemistry.org
- Definition and example – Chem.ucla.edu
- Definition – Chemtube3d.com
- Definition and mechanism – Name-reaction.com
- Definition and mechanism – Chem.libretexts.org
- Example and Mechanism – Chem.ucalgary.ca