Balz-Schiemann Reaction
Definition: What is Balz Schiemann Reaction?
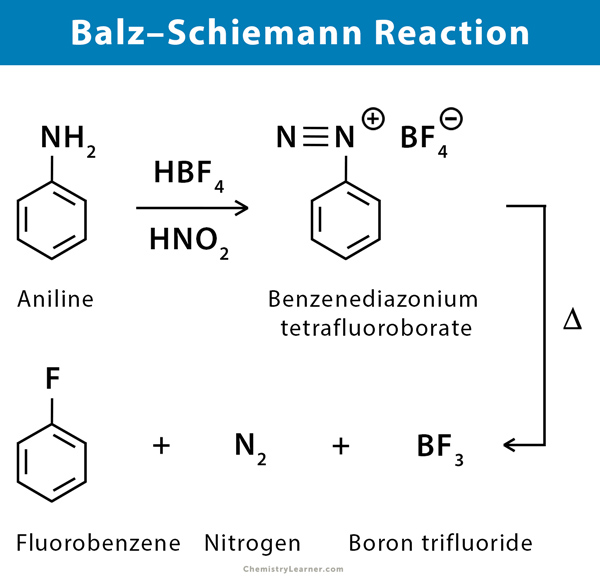
The Balz–Schiemann reaction is a chemical reaction in which a primary aromatic amine is transformed into an aryl fluoride via diazonium tetrafluoroborate intermediate. The primary reactants are aryl amines, nitrous acid, and fluoroboric acid. The process involves the conversion of arylamines to aryl fluorides via diazotization and subsequent thermal decomposition of the derived tetrafluoroborates or hexafluorophosphates. The decomposition may also be induced photochemically [1-2].
This reaction is especially vital for synthesizing the specifically fluorinated aromatic compounds, of which the decomposition in the absence of solvent usually affords a reasonably good yield of aromatic fluorides. The reaction is similar to the Sandmeyer reaction, which converts diazonium salts to other aryl halides.
The reaction is named after the German chemists Günther Schiemann and Günther Balz, who reported the reaction in a 1927 journal.
Example of Balz Schiemann Reaction
An example of Balz–Schiemann reaction is the conversion of phenylamine (aniline) into phenyl fluoride (fluorobenzene) using nitrous acid, fluoroboric acid, and heat [1-3].
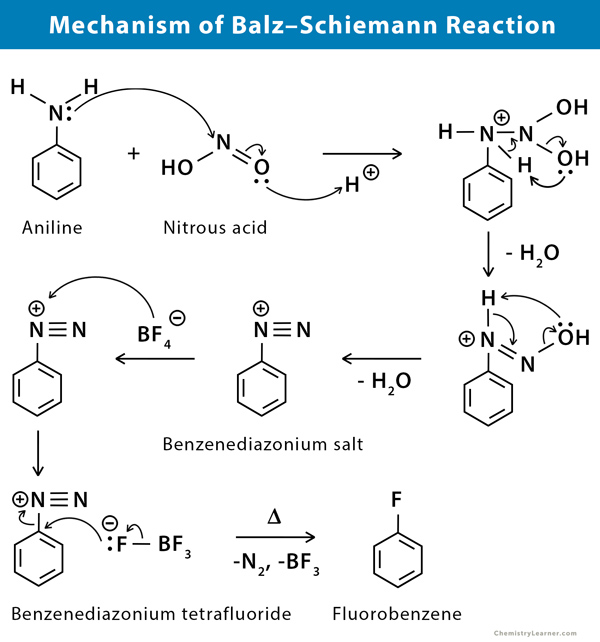
Mechanism of Balz Schiemann Reaction
While the Balz–Schiemann reaction is thought to proceed via an SN1 reaction mechanism, reactivity trends have been unpredictable. Aromatic amines undergo diazotization under the influence of nitrous acid. Fluoroboric acid is added to give rise to the corresponding aryl diazonium salt. This aryl diazonium salt is subjected to heat to undergo thermal decomposition and give the aryl fluoride along with nitrogen gas and boron trifluoride [1-3].
References
- Review – Researchportal.bath.ac.uk
- Review – Onlinelibrary.wiley.com
- Example and mechanism – Scientificupdate.com