Birch Reduction
Definition: What is Birch Reduction?
Birch reduction is an organic redox reaction that is used to convert aromatic compounds into dienes. The reaction is carried out by sodium or potassium metal dissolved in liquid ammonia in the presence of alcohol. Two hydrogen atoms are added at the opposite ends of the molecule at 1,4 positions. Birds reaction can be applied in the total synthesis of natural products [1-3].
The history of this reaction goes back to 1944 when Australian chemist Arthur Birch reported its findings.
Examples of Birch Reduction
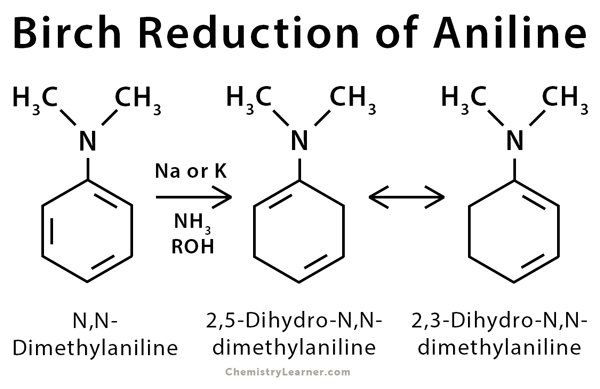
Birch reduction takes place in naphthalene and aniline. N, N-dimethylaniline was one of the earliest substrates used by Birch [4-5].
Mechanism of Birch Reduction
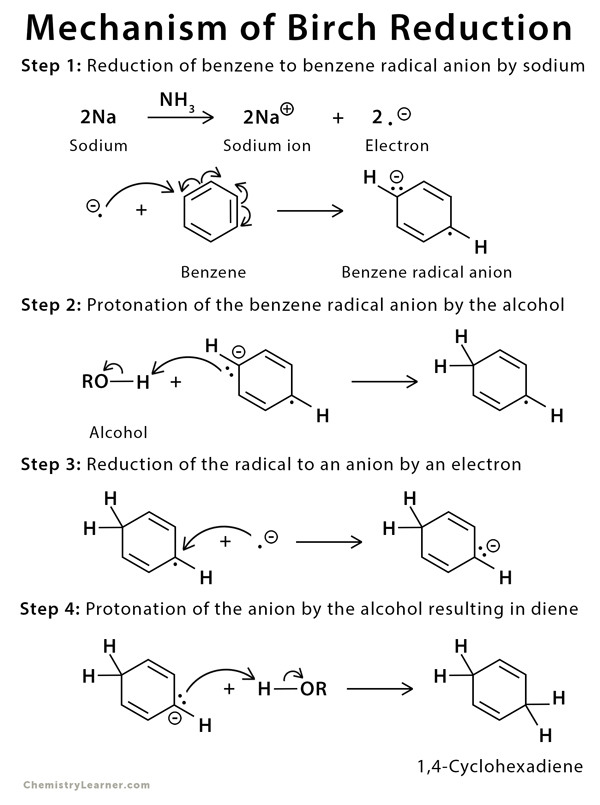
The Birch reduction of benzene takes place through a single electron transfer from sodium metal to the benzene ring to form a radical anion. The anion is protonated by alcohol. This process is repeated twice until two hydrogen atoms are added to the ring [6-7].
References
- Definition – Name-reaction.com
- Definition – Organic-chemistry.org
- Definition – Researchgate.net
- Example – Nature.com
- Example – Research.cm.utexas.edu
- Mechanism – Chemgapedia.de
- Mechanism – Chem.libretexts.org







I’m thankful, it helps me a lot in exam preparation,
Can you suggest me some articles for recent discoveries