Boyle’s Law
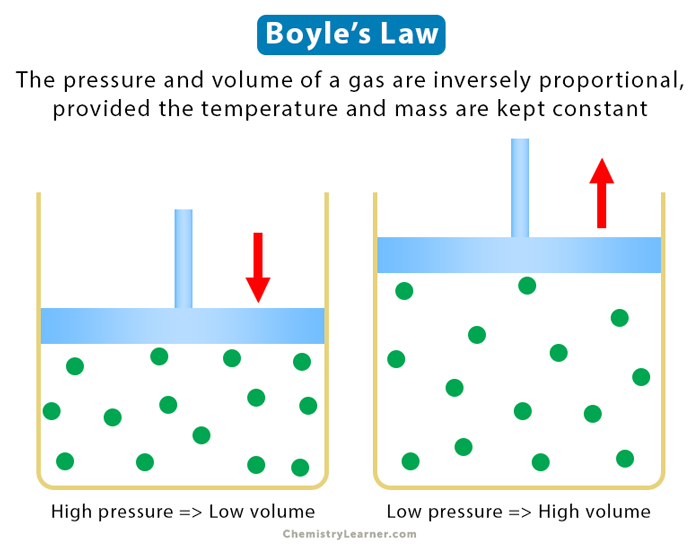
Boyle’s law is an experimental gas law that explains the relationship between pressure and volume. According to Boyle’s law, the pressure and volume are inversely proportional, provided the temperature and mass remain unchanged [1-4].
The law was named after Anglo-Irish physicist and chemist Robert Boyle, who published the law in 1662.
Equation
Suppose P is the pressure and V is the volume of the gas. Mathematically, Boyle’s law is given by [1-6]
P ∝ 1/V
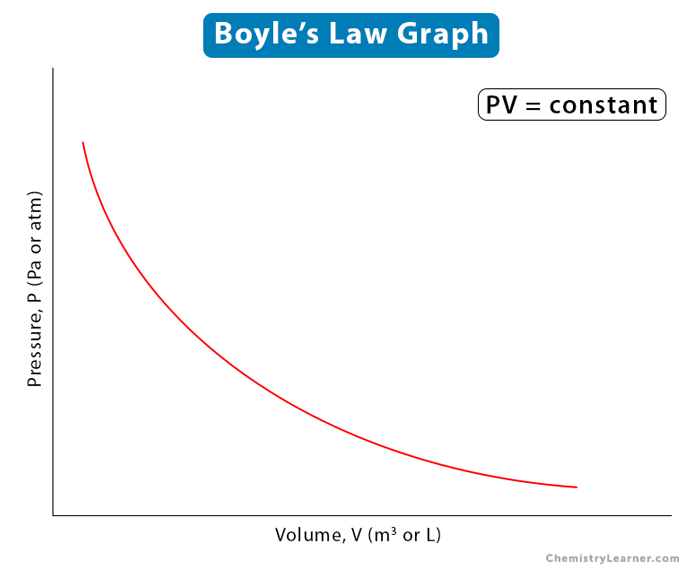
Or, P = k/V
Or, PV = k
Where
P : Pressure
V : Volume
k : Proportionality constant
This equation states that the product of pressure and volume is a constant for a given mass confined to a container as long as the temperature remains unchanged. A graphical representation of the above equation is shown below.
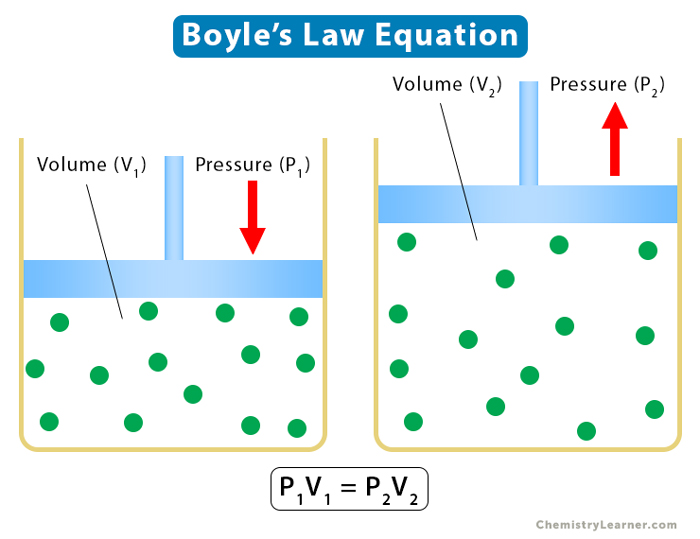
Boyle’s law can be used to establish a relationship between two states of a gas. Suppose the gas with pressure P1 and volume V1 expands or shrinks to pressure P2 and volume V2. Then, using Boyle’s law equation,
P1V1 = k and P2V2 = k
From the above two equations
P1V1 = P2V2
This equation shows that as the pressure increases, the volume decreases and vice versa. For example, when the pressure doubles, the volume is decreased by half. Also, the units of pressure and volume must be consistent. P1 and P2 must be expressed in Pa or atm. V1 and V2 must be expressed in m3 or L.
Examples
Here are some examples of Boyle’s law in real life [3].
Respiration and Breathing: When we inhale, our lungs expand. As a result, the volume increases, and pressure decreases. The air pressure inside the lungs is less than the environmental pressure. Hence, oxygen from the environment fills up the lungs. The reverse happens during exhalation. When we exhale, the lungs shrink. Its volume decreases, and pressure increases. The air pressure inside the lungs is higher than the environmental pressure. Hence, the air is expelled out of the lungs.
Syringe: A syringe draws liquid from a small vial and injects it into a body. It consists of a barrel, needle, and plunger. When the plunger is pulled, the volume inside the barrel increases, resulting in a decrease in pressure. Any fluid flows from high pressure to a low-pressure region. Since the pressure outside the syringe, near the needle, is higher than the pressure inside it, fluid will flow into the syringe. The reverse also holds. When the plunger is pushed, it creates a low volume and high pressure inside the syringe. As the pressure inside is higher than outside, the fluid will flow out of the syringe.
Balloon: When a balloon filled with air is squeezed, its volume decreases, and pressure increases. If the balloon is squeezed further, the pressure inside the balloon is so high that it causes the balloon to burst.
Problems and Solutions
Problem 1: A gas confined to a volume of 2 L at a pressure of 10 atm. It can flow into a 10 L container by opening the valve that connects the two containers. What is the final pressure of the gas?
Solution:
Given
P1 = 10 atm
V1 = 2 L
V2 = 10 L
From Boyle’s law,
P1V1 = P2V2
Or, P2 = P1V1/ V2
Or, P2 = 10 atm x 2 L/ 10 L
Or, P2 = 2 atm
Problem 2: A gas exerts a pressure of 5 kPa on the walls of a container. When the container is emptied into a 12 L container, the pressure exerted by the gas increases to 8 kPa. Find the volume of container 1.
Solution:
Given
P1 = 5 kPa
P2 = 8 kPa
V2 = 12 L
From Boyle’s law,
P1V1 = P2V2
Or, V1 = P2V2/P1
Or, V1 = 8 kPa x 12 L/5 kPa
Or, V1 = 19.2 L
FAQs
Ans. The variables involved in Boyle’s law are pressure and volume. On the other hand, temperature and the number of moles of gas are the constants.