Cannizzaro Reaction
Definition: What is Cannizzaro Reaction?
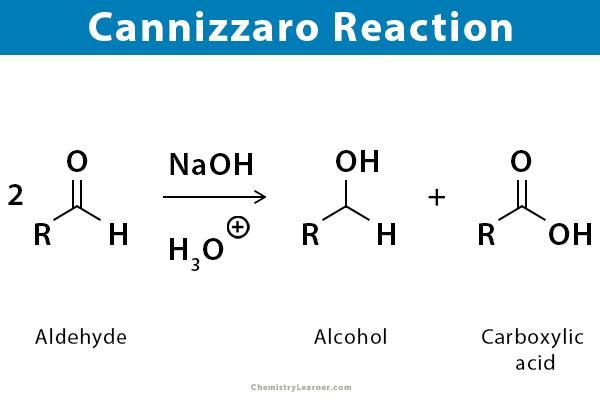
Cannizzaro reaction is an organic reaction of an aldehyde without active hydrogen that undergoes a redox reaction under the action of a strong base. Examples of aldehydes without active hydrogen include vanillin, benzaldehyde, syringaldehyde, and formaldehyde. They undergo intramolecular and intermolecular oxidation-reduction reactions under the action of a strong base (NaOH) to form a molecule of carboxylic acid and a molecule of alcohol [1-5].
The history of this reaction goes back to 1895 when Italian chemist Stanislao Cannizzaro obtained benzaldehyde and benzyl alcohol by treating benzaldehyde with grass ash.
Examples of Cannizzaro Reaction
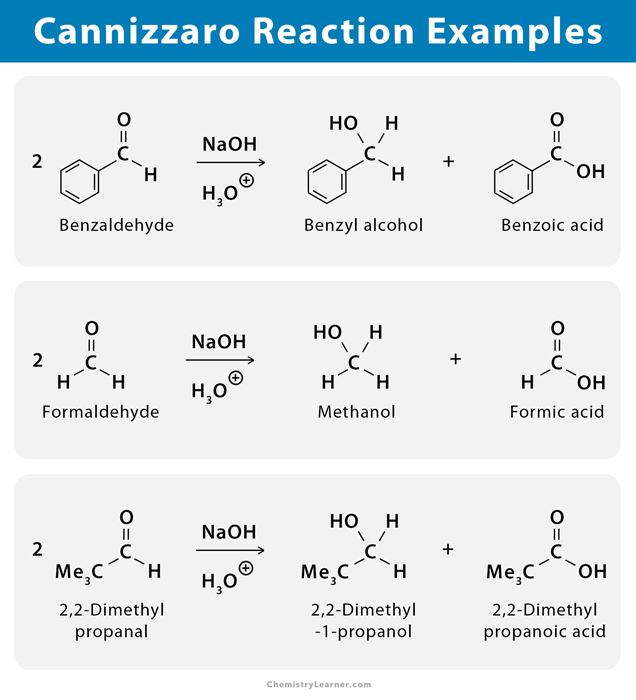
Benzaldehyde and formaldehyde can undergo Cannizzaro reaction, resulting in their corresponding alcohols and carboxylic acids [1-4].
Mechanism of Cannizzaro Reaction
The reaction begins with a hydroxide attack on the carbonyl carbon followed by deprotonation to give a dianion. The unstable dianion releases a hydride anion, which attacks another molecule of aldehyde. In this process, the dianion converts to a carboxylate anion and the aldehyde to an alkoxide. The alkoxide then picks up a proton from water to provide the final alcohol product. At the same time, the carboxylate is converted to the carboxylic acid product after acid workup [1-3,5].
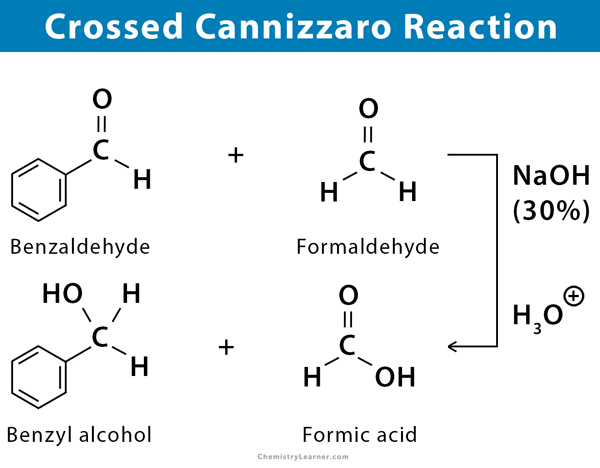
Crossed Cannizzaro Reaction
If an aldehyde without any α-hydrogen is made to react with formaldehyde in the presence of a base to give formic acid and an alcohol, it is called crossed Cannizzaro reaction. This reaction is quite useful for synthesizing a high yield of alcohol.
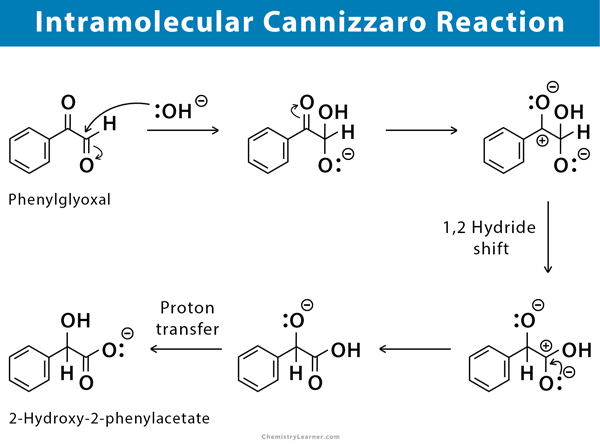
Intramolecular Cannizzaro Reaction
Intramolecular Cannizzaro reactions have been frequently studied as models for reactions occurring within an enzyme-substrate complex. Although several intramolecular Cannizzaro reactions have been reported, only phenylglyoxal has been investigated with details. Here, simultaneous oxidation and reduction take place at both the centers of the same molecule.
Uses and Applications of Cannizzaro Reaction in Industry
The combination of crossed Cannizzaro reaction and aldol condensation is used in the industry to prepare polyols. Polyols are very useful and find many industrial applications.
- Neopentyl glycol is used in polyesters for resins used in airplane or boat manufacturing, varnish coatings, synthetic lubricants, and plasticizers. The neopentyl structure provides excellent resistance to light, heat, and hydrolysis.
- Pentaerythritol is as a raw material in the varnish industry, while its tetranitrate is explosive. Some esters of pentaerythritol with higher fatty acids are being used as oil additives, plasticizers, and emulsifiers.
- Trimethylolpropane is used in several applications as a glycerin substitute in alky resins and for the preparation of polyesters and polyurethanes.
References
- Definition, example, and mechanism – Chem.libretexts.org
- Definition, examples, and mechanism – Onlinelibrary.wiley.com
- Definition, example, and mechanism – Chemistryscore.com
- Definition and example – Chemistry.msu.edu
- Definition and mechanism – Name-reaction.com