London Dispersion Forces
Table of Contents
What are London Dispersion Forces
London dispersion forces are weak, attractive intermolecular forces between two atoms or two nonpolar molecules due to temporary dipoles created by electrons’ motion. They belong to the category of van der Waals forces [1-7].
These forces are named after German physicist Fritz London, who first proposed their existence in 1930.
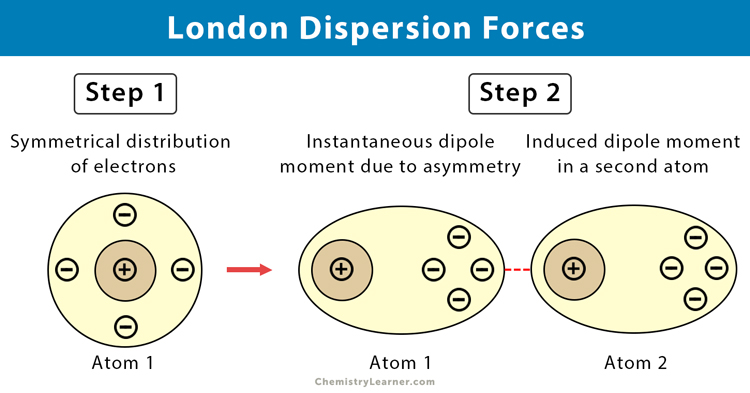
What Causes London Dispersion Forces [1-3]
An atom consists of a nucleus and electrons that move in orbits. At any time, the electrons can cluster around one part of the atom. The ease with which they move within the atom is called polarizability. When it happens, the ends of the atom become oppositely charged. This behavior of electrons results in an instantaneous dipole.
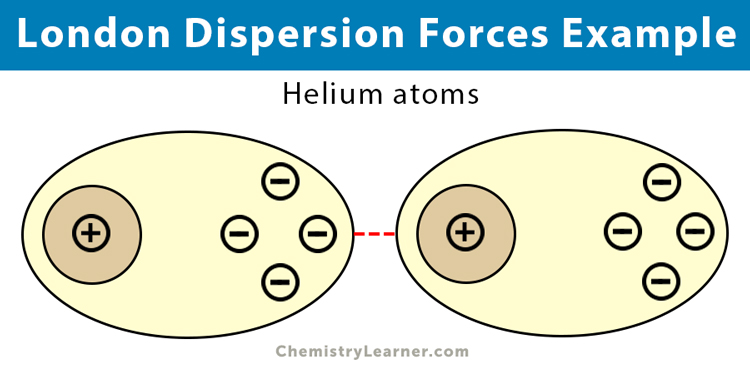
This weak and temporary dipole then influences neighboring atoms through electrostatic attraction and repulsion, thereby inducing dipoles. The induced dipoles are feebly attracted to one another through the dispersion forces. The strength of dispersion forces increases as the number of electrons in the atoms or nonpolar molecules increases.
When the positions of the electrons and protons flip, then the dipole reverses its polarity. These rapidly fluctuating dipoles are observed even in the most symmetrical molecule. It even occurs in noble gases, like neon, which consists of a single uncombined atom.
Properties of London Dispersive Forces
The following are facts and characteristics of the London dispersion forces [1]:
- Forces are attractive
- Forces are present between all atoms – polar and nonpolar
- Larger atoms display stronger forces than smaller ones, i.e., the strength increases down a particular group in the periodic table
- Easily polarizable atoms and molecules have stronger forces than harder polarizable ones
- Cylindrically shaped molecules allow more molecules to come close to one another than spherical ones, thereby increasing their strength
Examples of London Dispersive Forces
London dispersion forces are observed in nonpolar molecules. These include:
- Halogens: fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2)
- Nobel gases: helium (He), neon (Ne), argon (Ar), and krypton (Kr)
- Gases: oxygen (O2), nitrogen (N2), carbon dioxide (CO2), and phosphine (PH3)
- Hydrocarbons: methane (CH4), hexane (C6H6), carbon tetrachloride (CCl4), and ethane
Application of London Dispersion Forces
Intermolecular forces are important because they determine the physical properties of molecules like their melting point, boiling point, density, and enthalpies of fusion and vaporization. London dispersion forces are significant in both adhesion and sintering. During sintering, the detailed shape of the object at the crack tip and sintering neck is controlled by the dispersion forces [8].
Difference Between Dipole-dipole and London Dispersion
| Dipole-dipole Forces | London Dispersion Forces | |
|---|---|---|
| Definition | Occurs between two polar molecules | Occurs due to the presence of temporary dipole moments caused by moving electrons |
| Strength | Stronger than dispersion forces | Weaker than dipole forces |
| Polarity | Permanent | Temporary |
| Examples | HCl, H2O, and acetone | He, Cl2, and CCl4 |