Polarity
Polarity refers to the condition in which the electric charges on a molecule are separated, leading to a partial positive charge at one end and a partial negative charge at the other. It can also refer to the distribution of charges over a pair of atoms chemically bonded in a molecule. The former is known as molecular polarity, and the latter is called bond polarity. Polarity is a physical property of a compound that determines its solubility, melting point, and boiling point [1-4].
What Causes Polarity
Polarity comes from the uneven distribution of electrons among the various atoms in a molecule. The sharing of electrons in a chemical bond depends on the electronegativity values of the bonded atoms. More electronegative atoms such as oxygen, nitrogen, fluorine, and chlorine tend to attract bonded electron pairs and acquire partial negative charges. On the other hand, less electronegative atoms like hydrogen, carbon, and sulfur tend to acquire partial positive charges or remain neutral [1-4].
Electrons are unequally shared when two atoms with unequal electronegativities are bonded. The electron density increases in one atom and decreases in the other. As a result, partial charges develop in each atom. This charge separation leads to the formation of an electric dipole.
Because the amount of charge separated is less than one fundamental charge, they are called partial charges. They are denoted by the Greek symbol delta (δ) – δ+ for a partial positive charge and δ- for a partial negative charge. The product of the charge and the separation distance gives the bond’s dipole moment.
How to Determine Polarity in a Molecule
Since polar bonds are treated as vectors, the vector sum of all the bond polarities determines the polarity of a molecule. Moreover, the molecular shape affects polarity to a large extent. If the shape is such that the vectors cancel, the molecule has zero polarity and is said to be nonpolar. If the vectors do not cancel, the molecule has a nonzero polarity and is said to be polar [1-4].
Examples [1-4]
Polar Molecule
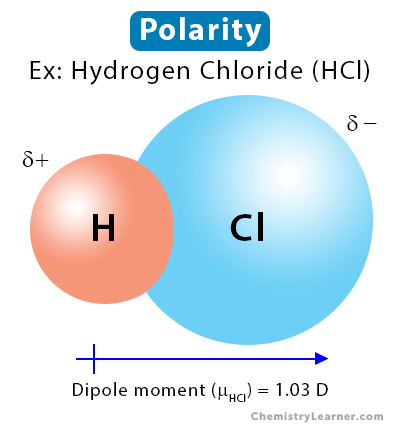
Hydrogen chloride (HCl) is an example of a polar molecule. Hydrogen (H) and chlorine (Cl) each share one electron to form a single covalent bond. Chlorine is more electronegative than hydrogen. Hence, it will pull the shared electron pair toward itself and acquire a partial negative charge.
On the other hand, there will be a deficiency of electrons on the hydrogen atom, resulting in a partial positive charge. The presence of positive and negative partial charges leads to polarity in HCl. The polarity is quantified by dipole moment, whose value for HCl is 1.03 D.
Nonpolar Molecule
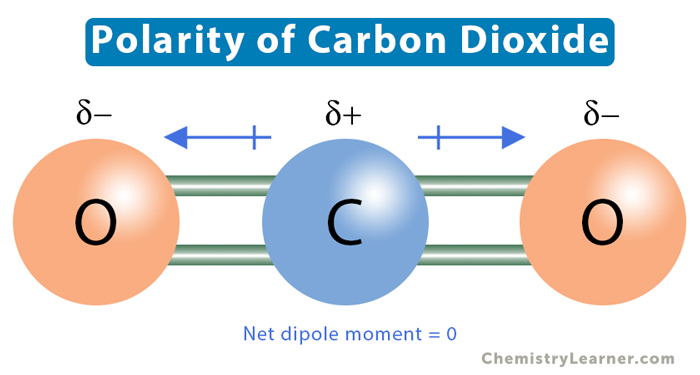
Carbon dioxide (CO2) is an example of a nonpolar molecule. CO2 has a linear structure, which means that carbon (C) and the two oxygen (O) atoms align in a straight line. There is a significant electronegativity difference between carbon and oxygen. The carbon atom forms a double covalent bond with each oxygen atom, resulting in two dipole moments. However, the two moments are equal in magnitude and opposite in direction. They will cancel, resulting in a net zero dipole moment.
Polarity of Some Common Compounds
FAQs
Ans. Polarity increases with the increasing s character of a covalent bond. Since a double bond has 33% s character compared to 25% in a single bond, its polarity increases.
Ans. Polar molecules have high intramolecular forces. As a result, polar compounds display high surface tension.
Ans. Generally, polar substances dissolve in polar liquids, and nonpolar substances dissolve in nonpolar liquids.