Chemical Equilibrium
Table of Contents
What is Chemical Equilibrium
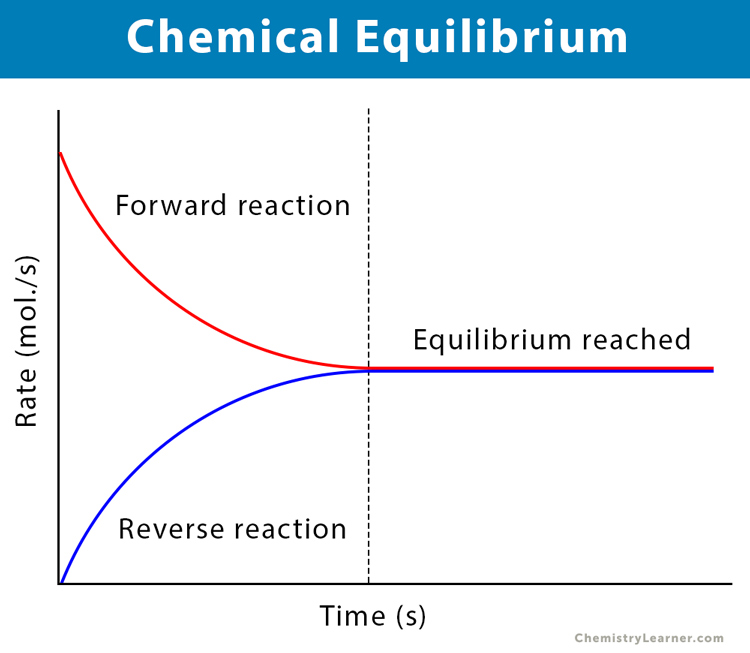
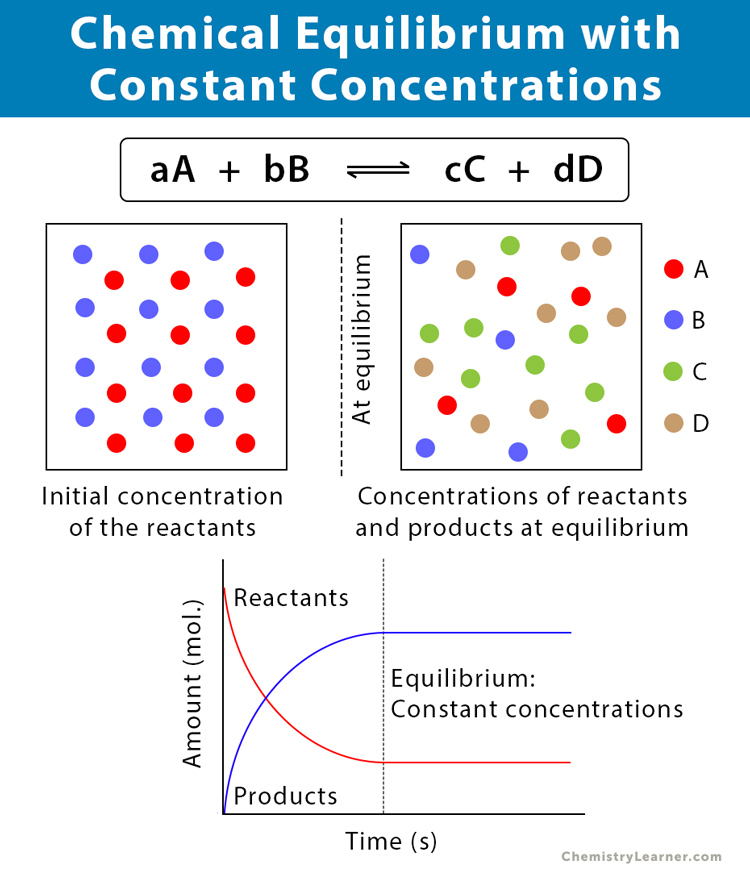
Chemical equilibrium is the condition of a system where the concentrations of the reactants and products do not change with time. Also, the system does not show any change in its properties.
During a reversible reaction, when the forward and reverse reaction rates become equal, the system achieves the state of chemical equilibrium. There is no further change in the concentrations of the reactants and the products.
Why is Chemical Equilibrium called Dynamic Equilibrium
During equilibrium, the forward reaction rate becomes equal to the backward reaction rate. At this time, the number of reactant molecules transforming into products and the number of product molecules converting into reactants are the same. This equilibrium state can be carried out with the same reactants anywhere with similar conditions.
Hence, chemical equilibrium is called dynamic equilibrium; as both the forward and reverse reactions do not cease, they continue to occur at equal rates. So, the ratio between reactants and products remains unchanged over time.
Principles of Chemical Equilibrium
The characteristics of chemical equilibrium are listed below:
- Equilibrium can be attained if the reversible reaction is carried out in a closed vessel.
- At equilibrium, both reactants and products are present.
- Both forward and reverse reactions proceed in opposite directions with equal rates.
- The state of equilibrium can be approached from either direction.
- The concentration of reactants and products does not change, along with its temperature, pressure, and color.
- According to Le Chatelier’s principle, by bringing a change in the observable properties of the system, such as temperature, pressure, or concentration, the equilibrium point can be shifted to the right or left side as required.
- A catalyst can hasten the approach but does not affect the position of the equilibrium.
Chemical Equilibrium Equation
Now, to understand the chemical equilibrium better, let us take an example of a reversible equation, where A and B are reacting to produce C and D and vice versa.
A + B ⇌ C + D
When the reactants react to synthesize products, it is known as a forward reaction. For example, A + B → C + D is showing a forward reaction.
On the other hand, when the products react among themselves to produce the reactants again, it is called a reverse reaction. For example, C + D → A + B is showing a reverse reaction.
Chemical Equilibrium Constant
Before proceeding, let us learn about the law of mass action. It states that the rate of a chemical reaction is directly proportional to the products of active masses of reactants, with each active mass term raised to the power equal to its stoichiometric coefficient in the balanced equation.
Active mass can be defined as the molar concentration of a substance actively taking part in a reaction.
Now, let us take a reversible reaction:
aA + bB ⇌ cC + dD
Following the law of mass action, the forward reaction rate (rf) ∝ [A]a [B]b, where [A] and [B] are the active masses or molar concentration of reactants A and B, and ‘a’ and ‘b’ are the stoichiometric coefficients of reactants A and B.
Therefore, rf = kf [A]a [B]b [the proportionality constant ‘k’ is known as the ‘rate constant’]
Similarly, the rate of reverse reaction (rr) can be written as:
rr = kr [C]c [D]d
We know, at equilibrium, the rate of forward and reverse reactions become equal, i.e., rf = rr.
Therefore,
kf [A]a [B]b = kr [C]c [D]d
or, kf/kr = [C]c [D]d / [A]a [B]b
or, kc =[C]c [D]d / [A]a [B]b
Here, kc = kf/kr is known as the ‘equilibrium constant.’
Examples of Chemical Equilibrium
Some examples of chemical equilibrium in industrial use are as follows:
- Ammonia is prepared for industrial use by Haber’s process combining nitrogen (N2) with hydrogen (H2) under high pressure, using iron as a catalyst.
N2(g) + 3 H2(g) ⇌ 2NH3(g)
- The contact process is the modern industrial method of producing sulfuric acid, where the fundamental reaction comprises the oxidation of sulfur dioxide (SO2) into sulfur trioxide (SO3).
2 SO2(g) + O2 (g) ⇌ 2 SO3(g)
Apart from the industrial uses, there are many examples of chemical equilibrium in real and everyday life.
- A bottle of fizzy cold drink contains carbon dioxide (CO2) dissolved in the liquid, also in the space between the liquid and the cap. There is a constant movement of CO2 from the liquid to the gas phase and vice versa. However, if we look at the bottle, there is no change, as the system is in equilibrium.
CO2(g) + H2O(l) ⇌ H2CO3(aq)
- An example of equilibrium in the human body every day is the interconversion of hemoglobin (Hb) to oxyhemoglobin (HbO2) and vice versa.
Hb(aq) + O2(g) ⇌ HbO2(aq)
Factors Affecting Chemical Equilibrium
According to the law of chemical equilibrium, i.e., Le Chatelier’s principle, ‘whenever a system at equilibrium is subjected to stress, the system will shift to relieve the stress.’
In simple words, if any of the factors affecting the equilibrium condition, like changes in temperature, pressure, and concentration, the system counteracts to regain the stable state.
Some important factors affecting the chemical equilibrium are discussed below:
1. Change in Concentration: The change in concentration of the reactants or products is relieved by the reaction.
According to Le Chatelier’s principle, adding more reactants to a system will shift its equilibrium to the right, i.e., the forward reaction rate will increase. In the same way, reducing the concentration of any product will shift the equilibrium to the right.
On the contrary, if more products get added to a system, the equilibrium will shift to the left to produce more reactants, i.e., the reverse reaction rate will increase. Also, when reactants are removed from the system, equilibrium shifts to the left.
2. Change in Pressure: If there is a change in pressure or volume, the system tries to restore the equilibrium by creating more or fewer moles of gas. As we know, the pressure and volume of a gas are inversely proportional to each other. If the pressure in a system decreases or the volume increases, the equilibrium shifts accordingly to the side of the reaction that involves the production of additional moles of gas. Similarly, if the volume of a system decreases or the pressure increases, fewer moles of gas will be favored.
3. Change in Temperature: The effect of temperature on chemical equilibrium depends upon the heat of the reaction (ΔH). Along with the equilibrium constant, the reaction rate is also affected by the change in temperature.
As per Le Chatelier’s principle, when the temperature increases in the case of exothermic reactions, the equilibrium shifts towards the right, i.e., to the reactant side. On the other hand, for endothermic reactions, the equilibrium shifts towards the product side with an increase in temperature.
4. Effect of a Catalyst: Catalysts speed up the reaction but do not affect the equilibrium position. It equally speeds up the forward and the reverse reaction, resulting in the reaction reaching its equilibrium faster. As a result, the same amount of reactants and products remain at equilibrium whether the reaction is catalyzed or non-catalyzed.
5. Effect of Addition of an Inert Gas: When inert gases like argon, krypton, and neon are added to a constant volume, they do not take part in the reaction. So, the equilibrium remains undisturbed.
Uses and Applications of Chemical Equilibrium
As mentioned, Le Chatelier’s principle of equilibrium involves parameters like temperature, pressure, and concentration of reaction, where a change in even a single parameter changes the equilibrium, leading to undesired product formation. So, in industry, the maintenance of optimal parameters helps in achieving the desired product. As we know, the concentration of both reactant and product plays a significant role in equilibrium. For instance, if the reactant concentration is higher, it will move towards a forward reaction. Similarly, a higher product concentration leads to a reverse reaction.
Hence, the main application of chemical equilibrium in industrial processes is to maximize the desired product concentration by minimizing the leftover reactants.
Summary and Conclusion
We have learned about chemical equilibrium, its characteristics, and the factors affecting it. Summing it all in the following points:
- A reversible reaction achieves equilibrium when its forward and backward reaction rates become equal.
- The ratio of rate constants of both forward and reverse reactions is called equilibrium constant ‘kc’.
- According to Le Chatelier’s principle, any change in concentration, temperature, or pressure of the reaction shift the reaction either left or right, yielding more products or reactants accordingly.
- Chemical equilibrium is of great importance in industrial applications, producing more desirable products, and enhancing profitability.
FAQs
Ans. No, the chemical reactions are not in equilibrium within living cells. They are open systems, with energy and matter entering and leaving continuously.
Ans. When any of the prevailing chemical equilibrium conditions are changed, the reaction tries to restabilize itself by increasing the forward or reverse reaction rate. Hence, the chemical equilibrium either shifts to the right or left.
References
- Chemical Equilibrium – Sciencedirect.com
- Chemical Equilibrium – Ncbi.nlm.nih.gov
- Factors that Affect Chemical Equilibrium – Courses.lumenlearning.com