Decomposition Reaction
What is a Decomposition Reaction?
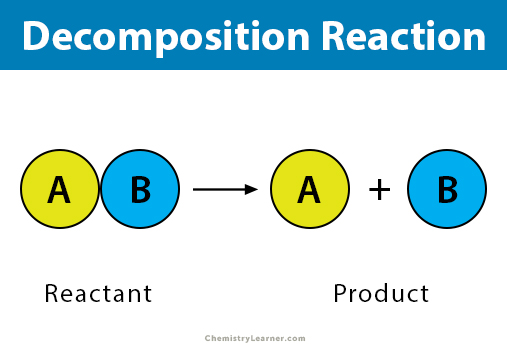
A decomposition reaction is a type of chemical reaction in which a compound breaks down into two or more substances. It is the opposite of a combination reaction. Sometimes, a complex chemical species breaks down into simple parts. The starting substance is called the reactant, and the resulting substances are called products [1-5].
General Equation
The following equation represents a general decomposition reaction.
AB → A + B
When writing an actual reaction, the reaction must be balanced.
What Occurs During a Decomposition Reaction?
During a decomposition reaction, the bonds between the atoms break down in the starting substance. The atoms then rearrange to form new bonds, resulting in new substances with properties different from the starting material.
Characteristics of Decomposition Reaction
- One single reactant and two or more products
- Requires energy
Examples of Decomposition Reaction
An example of a decomposition reaction is the breakdown of carbonic acid (H2CO3) to carbon dioxide (CO2) and water (H2O) [2].
H2CO3 (aq.) → CO2 (g) + H2O (l)
This reaction is significant and is responsible for the fizz in soda. Below are some more examples:
How to Balance Decomposition Reaction?
Let us take the example of the breakdown of potassium chlorate (KClO3) into potassium chloride (KCl) and oxygen (O2).
KClO3 (s) → KCl (s) + O2 (g)
This equation is unbalanced. In order to balance the equation, we inspect the number of oxygen (O) atoms on the right-hand side of the equation and compare it to the left. We notice that there are two oxygen atoms on the right and three on the left. So, we multiply the right-O by 3 and the left-O by 2. Note that we are going to multiply the entire compound KClO3 by 2 and balance the oxygen.
2 KClO3 (s) → KCl (s) + 3 O2 (g)
Now, we notice that the potassium (K) and chlorine (Cl) are not balanced. On the left-hand side of the equation, there are two atoms of K and Cl, whereas, on the right, there is only one atom of each. So, we multiply the compound KCl by 2 and obtain the final equation.
2 KClO3 (s) → 2 KCl (s) + 3 O2 (g)
Types of Decomposition Reaction
There are three types of decomposition reactions [1-3].
1. Thermal Decomposition
In this reaction, energy in the form of heat is required to break the bonds of the compound. The reaction is generally endothermic, i.e., heat is added to the reaction.
Rules for Thermal Decomposition
- Carbonates decompose into carbon dioxide and an oxide.
- Chlorates decompose into oxygen gas and a chloride.
- Hydroxides decompose into water and an oxide.
- Acids containing oxygen decompose into water and a molecular oxide.
- Oxides decompose into oxygen and another element.
Examples
- Calcium carbonate (CaCO3) decomposes into calcium oxide (CaO) and carbon dioxide (CO2) gas when heated.
CaCO3 (s) + heat → CaO (s) + CO2 (g)
- At high temperatures, magnesium carbonate (MgCO3) decomposes to magnesium oxide (MgO) and carbon dioxide (CO2).
CaCO3 (s) + heat → CaO (s) + CO2 (g)
- Thermal decomposition of phosphoric acid (H3PO4) produces diphosphoric acid (H4P2O7) and water (H2O).
2 H3PO4 → H4P2O7 + H2O
- Ammonium dichromate (NH4)2Cr2O7 decomposes on heating to produce nitrogen gas, water vapor, and solid chromium (III) oxide (Cr2O3) [6]
(NH4)2Cr2O7 (s) + heat → Cr2O3 (s) + N2 (g) + 4 H2O (g)
2. Electrolytic Decomposition
In this reaction, decomposition occurs when an electric current is passed through an aqueous solution of the compound.
Examples
- Electrolysis of water (H2O) to give hydrogen (H2) and oxygen (O2) gases
2 H2O (l) → 2 H2 (g) + O2 (g)
- Electrolysis of sodium chloride (NaCl) solution gives molten sodium (Na) and chlorine (Cl2) gas
2 NaCl (l) → 2 Na (l) + Cl2 (g)
3. Photo or Photochemical Decomposition
In this reaction, light (photons) is used to decompose the compound.
Examples
- Silver chloride (AgCl) decomposes into silver (Ag) and chlorine (Cl2) in the presence of sunlight
2 AgCl (s) + sunlight → 2 Ag (s) + Cl2 (g)
- In the presence of light, hydrogen peroxide (H2O2) decomposes into water (H2O) and oxygen (O2).
2 H2O2 (l) → 2 H2O (l) + O2 (g)
Another Type of Decomposition Reaction
Catalytic Decomposition
In this type of decomposition, the reaction occurs with the aid of a catalyst.
Example
- The decomposition of hydrogen peroxide (H2O2) to water (H2O) and oxygen (O2) is catalyzed by an enzyme called catalase [7].
2 H2O2 (l) + catalase (enzyme) → 2 H2O (l) + O2 (g)
Decomposition Reaction Examples in Real Life
The decomposition reaction has a few applications in the industry and daily life.
Industry
- Production of calcium oxide or quicklime
- Production of lithium oxide
- Preparation of oxygen and carbon dioxide
- In metallurgy, for the extraction of metals from their oxides and chlorides through electrolytic decomposition
Daily life
- When a soda bottle is opened, carbonic acid breaks down to produce water and carbon dioxide, which causes the fizz.
- During the digestion of food in our body, carbohydrates, fats, and proteins decompose to form many simpler substances.
- Photographic films have a coating of silver bromide, which on exposure to light splits into silver and bromine.
- When hydrogen peroxide is applied onto a cut or wound, the peroxide decomposes and forms oxygen bubbles and bursts.
- While baking a cake with baking powder (sodium bicarbonate), decomposition leads to the formation of carbon dioxide and sodium hydroxide.
- Many organic decompositions are observed in food like fermentation and fouling. Rotting of food and vegetable peels produce excellent nutrients and manure for the soil.
Laboratory
- In several analytical techniques like mass spectrometry, gravimetric analysis, and thermogravimetric analysis
FAQs
Ans. Most decomposition reactions require energy either in the form of heat, light, or electricity. Absorption of energy causes the breaking of the bonds present in the reacting substance which decomposes to give the product.
Ans. In a combination reaction, two or more substances combine to form a single product. However, in decomposition reaction, a single substance breaks down to give two or more substances. Therefore, the two are opposite to each other. Generally, combination reactions are mostly exothermic, while decomposition reactions are endothermic.
Ans. No. Not all decomposition are redox reactions.
Ans. No. The decomposition of aspirin is a double-replacement reaction.