Precipitation Reaction
What is Precipitation Reaction?
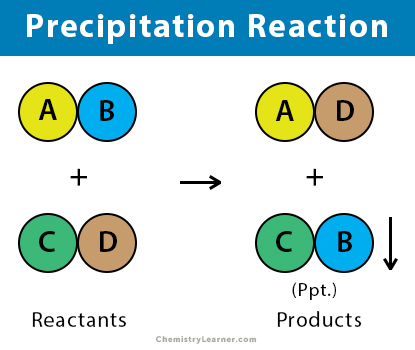
A precipitation reaction forms an insoluble salt when two soluble salts combine. The resulting solution is visibly cloudy due to the formation of small aggregation of the insoluble salt. This insoluble salt falls out of the solution and is filtered to form the precipitate. Precipitation reactions can help determine the presence of various ions in solution, most of which are formed from transition metals. This reaction is a double-replacement reaction [1-7].
General Equation
The general equation of the precipitation chemical reaction is given by:
AB + CD → AD + BC
Predicting Precipitation Reactions
The fundamental rule used to tell whether a precipitate will form or not is the solubility rule. According to this rule, if an ion is soluble, then it remains in its aqueous ion form. If an ion is insoluble, it forms a solid with an ion from the other reactant. If all the ions in a reaction are shown to be soluble, then no precipitation reaction occurs. The following table shows the solubility of various compounds. This table can be used to find and identify a precipitate [1,2].
Compounds soluble in water | Exception |
All compounds of Li+, Na+, K+, Rb+, Cs+, and NH4+ | None |
All compounds of NO3− and C2H3O2− | None |
Compounds of Cl−, Br−, and I− | Ag+, Hg22+, and Pb2+ |
Compounds of SO42- | Hg22+, Pb2+, Sr2+, and Ba2+ |
Compounds insoluble in water | Exception |
Compounds of CO32− and PO43− | Compounds of Li+, Na+, K+, Rb+, Cs+, and NH4+ |
Compounds of OH− | Compounds of Li+, Na+, K+, Rb+, Cs+, NH4+, Sr2+, and Ba2+ |
How to Balance Precipitation Reactions
Precipitation reaction can be balanced by ensuring that an equal number of atoms are present on both sides of the equation. Consider the following example:
Iron (III) chloride (FeCl3) and sodium hydroxide (NaOH) react to form sodium chloride (NaCl) and insoluble iron (III) hydroxide (Fe(OH)3).
FeCl3 (aq.) + NaOH (aq.) → NaCl (aq.) + Fe(OH)3 (s/ppt.)
The equation is unbalanced. In order to balance the equation, we look at the Fe, Na, Cl, and OH–. We notice that there are three Cl atoms on the left-hand side of the equation and only one on the right. So, we multiply the compound NaCl by 3 and obtain the following:
FeCl3 (aq.) + NaOH (aq.) → 3 NaCl (aq.) + Fe(OH)3 (s/ppt.)
Next, we notice that Na is unbalanced, as there are three atoms on the right and only one on the left. So, we multiply NaOH by 3 and obtain:
FeCl3 (aq.) + 3 NaOH (aq.) → 3 NaCl (aq.) + Fe(OH)3 (s/ppt.)
Examples of Precipitation Reaction
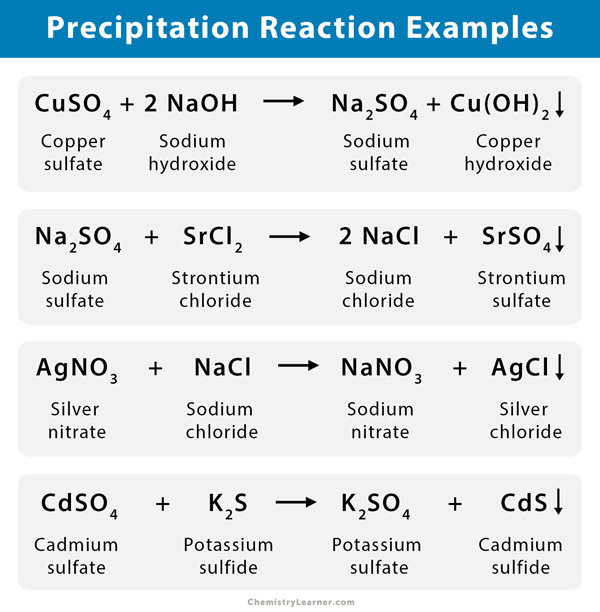
Here are some examples of precipitation reactions between salts of metals dissolved in aqueous solution [1-7].
- Lead nitrate (Pb(NO3)2) and potassium iodide (KI) react to form potassium nitrate (KNO3) and insoluble lead iodide (PbI2).
Pb(NO3)2 (aq.) + 2 KI (aq.) → 2 KNO3 (aq.) + PbI2 (s/ppt.)
- Barium chloride (BaCl2) reacts with sodium sulfate (Na2SO4) to form sodium chloride (NaCl) and insoluble barium sulfate (BaSO4).
BaCl2 (aq.) + Na2SO4 (aq.) → 2 NaCl (aq.) + BaSO4 (s/ppt.)
- Mixing solutions of calcium chloride (CaCl2) and sodium carbonate (Na2CO3) forms the precipitated calcium carbonate (CaCO3) along with sodium chloride (NaCl)
CaCl2 (aq.) + Na2CO3 (aq.) → 2 NaCl (aq.) + CaCO3 (s/ppt.)
- In the chemical reaction between potassium chloride (KCl) and silver nitrate (AgNO3), silver chloride (AgCl) is precipitated out, and potassium nitrate (KNO3) remains in solution.
KCl (aq.) + AgNO3 (aq.) → KNO3 (aq.) + AgCl (s/ppt.)
Precipitation Reaction Examples in Real Life
Here are some examples of precipitation reactions in everyday life [3,4]:
- Precipitation in water pipes: Hard water consists of many impurities. Magnesium and calcium oxides are generally found in water pipes as precipitates.
- Formation of kidney stone: The reaction between calcium ions and oxalic acid that are present in food results in calcium oxalate, which is a type of kidney stone.
- Formation of the immune complex: Soluble antigens combine with soluble antibodies in the presence of an electrolyte (solution) at a suitable temperature and pH to form an insoluble visible complex in the human body.
Practical Uses of Precipitation Reaction
One of the applications of precipitation reaction is determining whether a particular element is present in a solution. Here are some of its uses [3,4]:
- Wastewater treatment: A chemical is added to wastewater so that the pollutants and impurities (e.g., lead) precipitate out from the water, which is then sent for further purification.
- Metallurgy: Oxalic acid is mixed with seawater and brine water to extract calcium and magnesium, forming precipitates.