Reversible Reaction
What is Reversible Reaction
A reversible reaction is a chemical reaction in which the conversion of reactants to products and converting products to reactants co-occur. Therefore, in a reversible reaction, the reactants and the products are never fully consumed, nor is the reaction complete. Instead, the reaction will come to equilibrium if the concentrations of the reactants and products remain constant. An example of a reversible process in our daily lives is melting ice into water and freezing water into ice. [1-6].
General Equation for Reversible Reaction
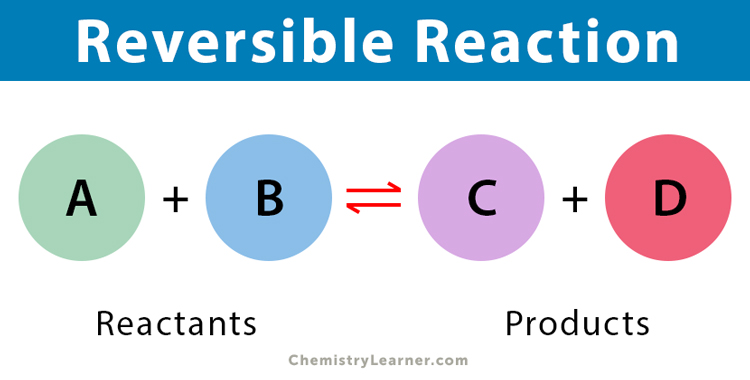
The general formula for a reversible reaction is given as follows:
A + B ⇋ C + D
Symbol
The symbol for a reversible reaction is a double-arrow: ⇋
How does a Reversible Reaction Work
In a reversible reaction, the molecules in a closed system collide with energies to break chemical bonds and form new products. However, the products are highly energetic, and their molecules also collide to break new bonds. As a result, new bonds form to give back the reactants.
Conditions for Reversible Reaction
Here are the conditions for a reversible reaction.
- It should occur in a closed container
- None of the products is allowed to form a precipitate
According to Le Chatelier’s principle, a reversible reaction is self-correcting. Suppose there is a change in concentration, temperature, or pressure. In that case, the system will naturally shift towards equilibrium.
Examples of Reversible Reaction
Here are a few examples of the reversible reaction [3-6].
1. Reaction between hydrogen (H2) and iodine (I2) to produce hydrogen iodide (HI).
H2 (g) + I2 (g) ⇋ 2 HI (g)
2. Nitrogen (N2) reacting with hydrogen (H2) to produce ammonia (NH3).
N2 (g) + 3 H2 (g) ⇋ 2 NH3 (g)
3. Sulfur dioxide (SO2) reacts with oxygen (O2) to make sulfur trioxide (SO3)
2 SO2 (g) + O2 (g) ⇋ 2 SO3 (g)
The forward reaction is exothermic, and the reverse reaction is endothermic.
4. White solid of ammonium chloride (NH4Cl) breaks down in ammonia (NH3) and hydrogen chloride (HCl) when heated.
NH4Cl (s) ⇋ NH3 (g) + HCl (g)
5. Blue hydrated copper sulfate (CuSO4.5H2O) converts into white anhydrous copper sulfate (CuSO4) when heated.
CuSO4.5H2O (s) ⇋ CuSO4 (s) + 5 H2O (g)
The forward reaction is endothermic, and the reverse reaction is exothermic.
Reversible and Irreversible Reaction
A chemical reaction in which the entire reactants are converted into products is called an irreversible reaction. Unlike a reversible reaction, an irreversible reaction proceeds only in one direction. The reaction is complete and is indicated by a single arrow: →
An example of an irreversible reaction is the combustion of fuel, like ethanol (C2H5OH), giving rise to carbon dioxide (CO2) and water (H2O).
C2H5OH (aq.) + 3 O2 (g) → 2 CO2 (g) + 3 H2O (g)