Paper Chromatography
Table of Contents
Paper chromatography is a simple and cost-effective separation technique that separates and identifies different components in a mixture. [1-4]
Principle
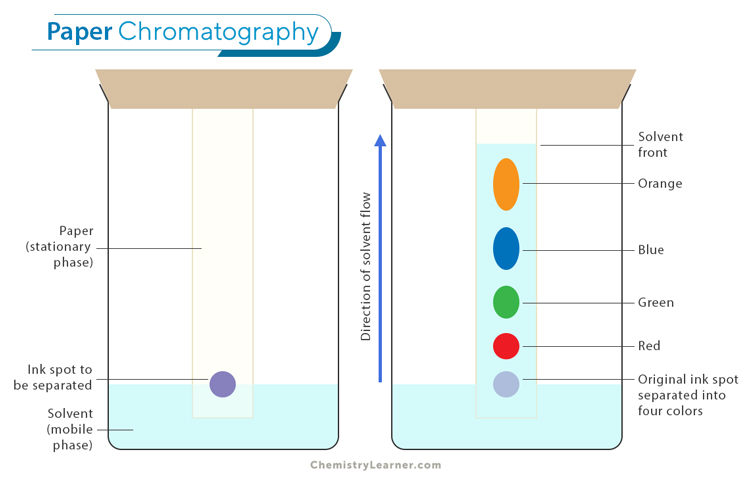
In paper chromatography, a specialized paper acts as the stationary phase, while a liquid solvent is the mobile phase. The mixture to be analyzed is applied to the paper. As the solvent moves up through capillary action, it carries along the individual components of the mixture at different rates based on their solubility and affinity for the stationary phase.
The principle behind paper chromatography lies in the differential partitioning of compounds between the stationary and mobile phases. The stationary phase typically consists of cellulose fibers embedded in filter paper or thin-layer chromatography plates. These fibers provide an adsorbent surface for compounds to interact with.
How Does Paper Chromatography Work
Understanding the mechanism behind paper chromatography requires knowledge of several key processes. [1-4]
The first process is capillary action, which refers to the ability of liquids to flow through narrow spaces against gravity. In paper chromatography, capillary action allows the solvent to move up the paper strip due to its attraction to the fibers in the paper. As the solvent moves up, it carries the solutes in the analyzed mixture. This migration of solutes is driven by two main mechanisms: adsorption and partitioning.
Adsorption occurs when solute molecules adhere to the fibers or other surfaces within the paper. It can be influenced by polarity and molecular size, with more polar or larger molecules having stronger interactions with the stationary phase.
Conversely, partitioning involves solute molecules distributing themselves between two immiscible phases – in this case, between the stationary phase (paper) and mobile phase (solvent). The extent of partitioning depends on factors such as solute polarity and affinity for either phase.
As solutes migrate up through capillary action, they may experience different degrees of adsorption and partitioning along their journey. This results in their separation based on their characteristics. By analyzing how far each component migrates on a chromatogram – a visual representation of separated components – scientists can determine properties such as retention factor (Rf) values and identify unknown substances based on known reference compounds.
Stationary and Mobile Phases
Stationary and mobile phases play crucial roles in separating components of a mixture. [1-4]
The stationary phase refers to the absorbent material fixed on the chromatography paper. It can be made of cellulose or other materials with high absorbency. The stationary phase acts as a substrate for the sample mixture to interact with during separation.
On the other hand, the mobile phase is the solvent or liquid that moves through the stationary phase, carrying the sample components. The mobile phase must have good solubility with the components of interest. It should be able to flow easily through the paper.
As the mobile phase moves through the stationary phase, it interacts differently with each mixture component based on their solubility and affinity for both phases. This differential interaction leads to separation as different components travel at different rates along the paper.
Choosing an appropriate combination of stationary and mobile phases is important for effective separation in paper chromatography. Factors such as polarity, viscosity, and compatibility between phases must be considered to achieve optimal results.
Paper Chromatography Experiment
Performing a paper chromatography experiment involves several essential steps to ensure accurate results. The process begins with preparing samples for paper chromatography, then spotting the sample on the paper strip, and finally, developing the chromatogram. [1-4]
Preparing the samples is crucial in obtaining reliable data. It involves selecting appropriate substances to analyze and ensuring they are suitable for chromatography. Samples can be liquid or solid and must be dissolved or crushed into a solution before application.
Next, spotting the sample on the paper strip is done carefully to ensure accurate separation. A small spot of the prepared sample is placed near one end of a designated area on the filter paper strip. It is essential to use a capillary tube or micropipette for precise and consistent application.
Once all samples are spotted on the filter paper strip, it is time for the development of the chromatogram. This step involves placing one end of the strip into a solvent traveling up through capillary action. The choice of solvent depends on factors such as solubility and desired separation distance.
As the solvent moves up through the filter paper strip, it carries different components in each sample. These components separate based on their affinity for stationary (filter paper) and mobile (solvent) phases. The separation occurs due to differences in molecular size, polarity, or other physical properties.
Throughout this process, it is important to maintain controlled conditions such as temperature and humidity to ensure reproducibility. Further analysis can be conducted once an optimal separation has been achieved, which can take several minutes or hours depending on various factors, including solvent choice and sample composition.
Applications
The diverse applications of paper chromatography across various fields are listed below. [1-4]
- It plays a crucial role in forensic analysis by separating and identifying different components in complex mixtures, such as blood or ink samples.
- Aids in the analysis of crime scene evidence, allowing forensic scientists to determine the presence of specific substances and identify potential suspects based on chromatographic patterns
- Enables the separation of different dyes used in food coloring, helping to ensure compliance with regulatory standards and quality control measures
- Determines the authenticity and safety of food products by identifying and quantifying specific components present in complex food matrices
- Separate and identify active ingredients, impurities, and by-products in pharmaceutical formulations.