Thin Layer Chromatography
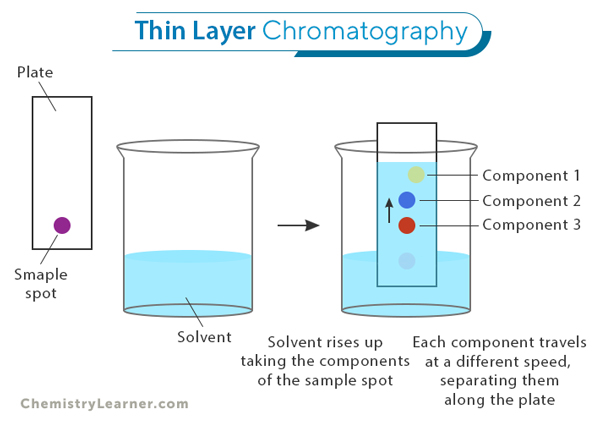
Thin Layer Chromatography (TLC) is a separation method that identifies and analyzes different components in a mixture. TLC involves placing a small amount of the sample to be analyzed onto a thin layer of adsorbent material, such as silica gel or alumina, which is coated on a glass or plastic plate. This plate is then developed by allowing a solvent to move up the plate through capillary action. [1-4]
Principle of Thin Layer Chromatography
The principle of Thin Layer Chromatography (TLC) is based on separating chemical compounds using stationary and mobile phases. [1-4]
A thin layer of adsorbent material, such as silica gel or alumina, is coated onto a solid support, usually a glass or plastic plate. This thin layer acts as the stationary phase. The sample mixture to be analyzed is spotted near the bottom of the plate.
The separation begins when the plate is placed in a developing chamber with a suitable solvent called the mobile phase. The mobile phase moves up through capillary action, carrying the sample components. Each component interacts differently with the stationary and mobile phases as it moves up the plate.
The interactions between the components and these two phases result in differential migration rates. Compounds with stronger interactions with the stationary phase will move more slowly than those with weaker interactions. It leads to the separation of individual components on the TLC plate.
Procedure for Thin Layer Chromatography
The first step in performing TLC is to prepare the sample. It involves dissolving or suspending the mixture of compounds in a suitable solvent to create a solution. The sample solution is then applied as a small spot near one end of a TLC plate using a capillary tube or micropipette. [1-4]
Next, the TLC plate needs to be prepared. It involves selecting an appropriate stationary phase (usually silica gel or alumina) and applying it evenly as a thin layer on a glass or plastic plate. The stationary phase should be activated by heating in an oven before use.
Once the sample spot has been applied to the TLC plate, it needs to be developed. This process involves placing the bottom edge of the plate into a developing chamber containing an appropriate mobile phase (a solvent or mixture of solvents). The mobile phase ascends through capillary action, carrying the components of the sample with it.
As the mobile phase moves up through the stationary phase, different compounds in the sample will separate based on their affinities for both phases. The separation occurs because some compounds interact more with the stationary or mobile phase than others.
After development, it is time to visualize and interpret the results. Visualization can be done by exposing the TLC plate to ultraviolet (UV) light or using chemical reagents that react with specific compounds on your plate. These visualization techniques help identify spots corresponding to different components in your sample.
Interpreting TLC results involves measuring how far each component has traveled relative to how far the solvent front has traveled. These distances can be estimated using the retention factor (Rf). It is calculated by dividing the distance traveled by the component by the distance traveled by the solvent front. Rf values can identify unknown compounds or compare results with known standards.
Applications of Thin Layer Chromatography
Thin Layer Chromatography (TLC) is a versatile analytical technique that finds applications in various fields. [1-4]
- Pharmaceutical Analysis: It is used for drug identification, purity testing, and quality control. It allows researchers to separate and analyze different components of pharmaceutical formulations, ensuring the safety and efficacy of drugs.
- Food Analysis: It is used to determine the presence of contaminants such as pesticides, mycotoxins, and food additives. It helps ensure that food products meet regulatory standards and are safe for consumption.
- Environmental Monitoring: It analyzes water samples for pollutants like heavy metals or organic compounds. This information aids in assessing environmental contamination levels and guiding appropriate remediation measures.
- Forensic Science: It assists forensic investigators in analyzing trace evidence such as fibers, dyes, or drugs found at crime scenes. By separating these substances into individual components using TLC, scientists can identify specific compounds and provide crucial evidence in criminal investigations.
Disadvantages of Thin Layer Chromatography
Despite its widespread use, thin layer chromatography has some limitations and disadvantages. One of the main drawbacks is its relatively low-resolution power compared to other chromatographic techniques. TLC is less effective in separating closely related compounds than High Performance Liquid Chromatography (HPLC) or Gas Chromatography (GC). It can be challenging to analyze complex mixtures accurately. [1-4]
Another limitation of TLC is its limited sample loading capacity. Due to the thin layer of stationary phase on the TLC plate, it can only accommodate a small sample. Therefore, it is less suitable for analyzing large volumes or concentrated samples.
Additionally, TLC can be more time-consuming compared to other chromatographic techniques. The separation process on the TLC plate may require more development time, especially when dealing with complex mixtures.