Claisen Rearrangement
Definition: What is Claisen Rearrangement?
A rearrangement is a reaction in which a molecule changes its bonds by transferring one atom or group from one position to another within the molecule. The Claisen rearrangement is an organic reaction in which an allyl vinyl ether or an allyl aryl ether undergoes a 3,3-sigmatropic rearrangement to produce a γ,δ-unsaturated carbonyl compound. This reaction is facilitated with the addition of heat or a Lewis acid. This reaction is an intramolecular, concerted process where bonds break and form at the same time. The Claisen rearrangement is essentially an irreversible process since the products are considerably more stable than the reactants, unlike the Cope rearrangement [1-6].
The history of this reaction goes back to 1912 when German chemist Rainer Ludwig Claisen discovered the thermally induced rearrangement of allyl phenyl ether.
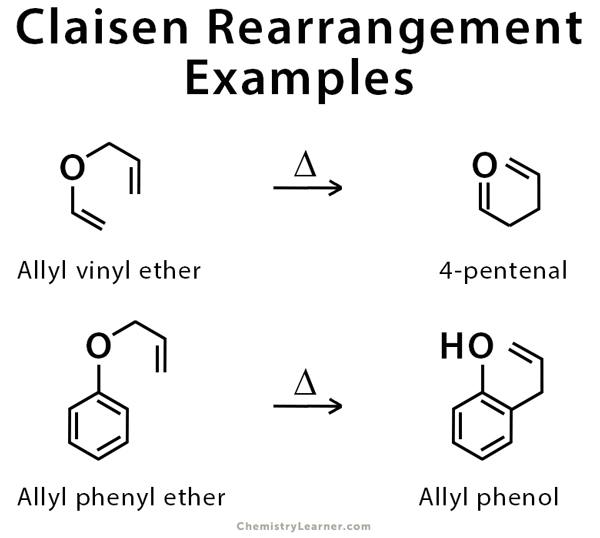
Examples of Claisen Rearrangement
This reaction is used for the rearrangement of allyl vinyl ether and allyl aryl ether like allyl phenyl ether. The latter is used to synthesize allyl phenol [3-5,7,8].
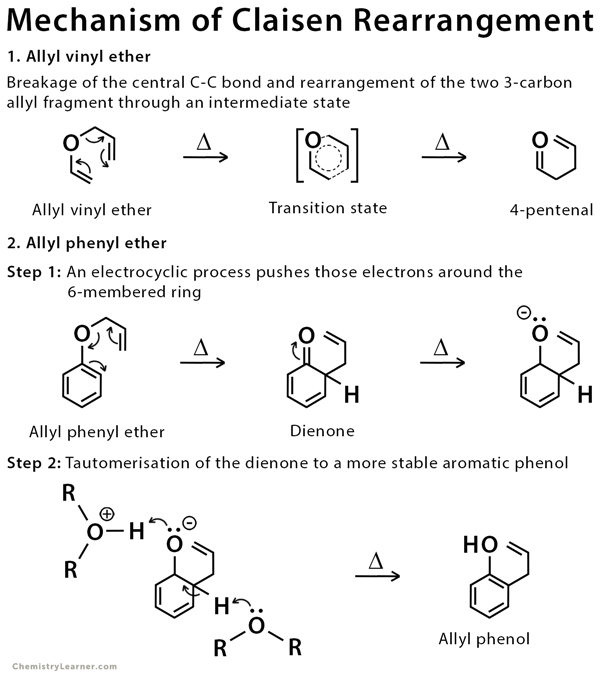
Mechanism of Claisen Rearrangement
The mechanism of this reaction closely resembles that of the Diels-Alder reaction. Proton tautomerism is expected in this reaction, where a proton is removed from one site in the molecule and placed at a different nearby site [4-7].
Uses and Applications of Claisen Rearrangement
The Claisen rearrangement is applied in nature for the rearrangement of chorismate to prephenate, a key intermediate in the synthesis of amino acids like phenylalanine and tyrosine.
References
- Definition – Chem.libretexts.org
- Definition – Chemtube3d.com
- Definition and example – Employees.csbsju.edu
- Definition, example, and mechanism – Ochempal.org
- Definition, example, and mechanism – En.chem-station.com
- Definion and mechanism – Name-reaction.com
- Example and mechanism – Chem.ucalgary.ca
- Examples – Chem.wisc.edu






I love this page…I learned mechanism very easily from this website…thank you