Clausius-Clapeyron Equation
The Clausius-Clapeyron equation is fundamental in thermodynamics and establishes a relationship between the vapor pressure of a substance to its temperature. It provides a valuable understanding of the behavior of substances undergoing phase changes, such as melting and boiling [1-4].
The equation is named after German physicist and mathematician Rudolf Clausius and French engineer and physicist Benoît Paul Émile Clapeyron. They independently derived the equation in the mid-19th century.
Formula and Plots
The Clausius-Clapeyron equation states that the logarithm of the vapor pressure of a substance is directly proportional to its reciprocal absolute temperature. According to it, the natural logarithm of the liquid’s vapor pressure (P) is determined by the molar enthalpy of vaporization of the liquid (ΔHvap), the ideal gas constant (R), and the temperature (T) of the system. Mathematically, the equation is represented by [4-6]
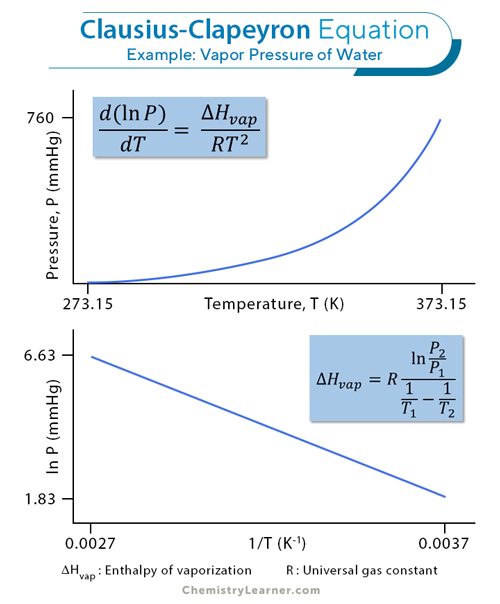
d (ln P)/ dT = ΔHvap/RT2
Integrating the above equation and assuming that ΔHvap does not depend on the system’s temperature, the Clausius-Clapeyron equation can be written in the following integrated form.
ln P = – ΔHvap/RT + C
Where C is an arbitrary constant. This equation can be rewritten as
P = A exp (- ΔHvap/RT)
Where A = exp (C). The plots of P vs. T and ln P vs. 1/T are shown below.
To eliminate C from the above equation, we consider two points 1 and 2 on the ln P vs. 1/T plot that give two values of the vapor pressure and temperature – (P1, T1) and (P2, T2). Plugging these values, we get
ln (P2/P1) = (ΔHvap/R) (1/T1 – 1/T2)
Rearranging the above equation, one can measure the molar enthalpy of vaporization of a liquid.
ΔHvap = R((ln P2 – ln P1)/(1/T1 – 1/T2))
Derivation
The Clausius-Clapeyron equation is derived from thermodynamics principles. A substance goes from phase 1 to phase 2 during a phase transition. Along the phase boundary, the two phases are in equilibrium. Their specific Gibbs free energies (g) are equal [1,7-9].
g1(p, T) = g2(p, T)
Suppose there is a change of temperature from T to T + dT. The corresponding change in pressure is from P to P + dP. The Gibbs free energy of phase 2 will change by the same amount as that of phase 1.
dg1 = dg2
Since the specific Gibbs energy is a function of temperature and pressure, we can write dg as follows:
dg = (∂g/∂T)P dT + (∂g/∂P)T dP
However,
(∂g/∂T)P = -s and (∂g/∂P)T = v
Such that,
dg = -sdT + vdP
Therefore, the equilibrium condition becomes
-s1dT + v1dP = -s2dT + v2dP
=> -(s1 – s2)dT + (v1 – v2)dP = 0
=> dP/dT = (s1 – s2)/(v1 – v2)
The entropy change associated with the phase transition is
(s1 – s2) = (h1 – h2)/T = Δh/T
Where Δh is the latent heat.
dP/dT = Δh/T(Δv)
The above expression is a generalized form of the Clausius-Clapeyron equation.
- When 1 = liquid and 2 = gas, Δh = ΔHvap is the latent heat of vaporization
- When 1 = solid and 2 = liquid, Δh = ΔHfus is the latent heat of fusion
- When 1 = solid and 2 = gas, Δh = ΔHsub is the latent heat of sublimation
Consider the liquid-gas phase transition, occurring at temperatures much lower than the critical temperature of the substance. The specific volume of the gas phase greatly exceeds that of the liquid.
vg >> vl and Δv = vg
Then,
dP/dT = ΔHvap/T(vg)
At low temperatures and pressure, one may approximate the gas to follow the ideal gas law so that
vg = RT/P
The Clausius-Clapeyron equation becomes
dP/dT = ΔHvap/T(RT/P)
=> d(ln P)/dT = ΔHvap/RT2
Uses and Applications
Applications of the Clausius-Clapeyron equation extend across diverse scientific disciplines, enabling one to make informed decisions regarding weather patterns, chemical reactions, and industrial processes involving phase changes [9-10].
- It helps meteorologists to understand cloud formation and precipitation patterns by relating temperature variations to changes in water vapor pressure.
- It aids chemists in determining reaction rates and equilibrium constants for reactions involving gases and volatile substances.
- It assists engineers in designing efficient heat exchangers and distillation systems by considering how temperature affects vapor pressures during separation processes.





This page is very well written and helpful. I’d like to remark that the CC equation is also very important in agriculture, it gets into the Penman formula used to compute potential evaporation of water from soils and crops, so that irrigation needs can be assessed on a physical basis.