Curtius Rearrangement
Definition: What is Curtius Rearrangement?
The Curtius rearrangement is a versatile reaction that converts a carboxylic azide to an isocyanate under thermal conditions with the loss of nitrogen gas. Isocyanates can be isolated during azide decomposition in inert solvents. By adding various nucleophiles, isocyanates can be transformed into other useful species [1-4].
The complete reaction sequence is called the Curtius Reaction. It is named after German chemist Theodor Curtius who reported it in two papers in 1890 and 1894.
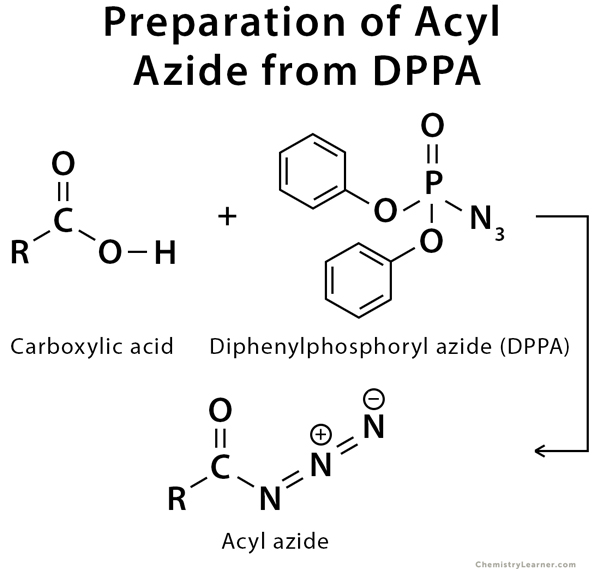
Preparation of Acyl Azide
The acyl azide is prepared by the direct reaction of a carboxylic acid with diphenylphosphoryl azide (DPPA).
Example of Curtius Rearrangement
The Curtius rearrangement can be used to convert phenyl azide into phenyl carbamate [2].
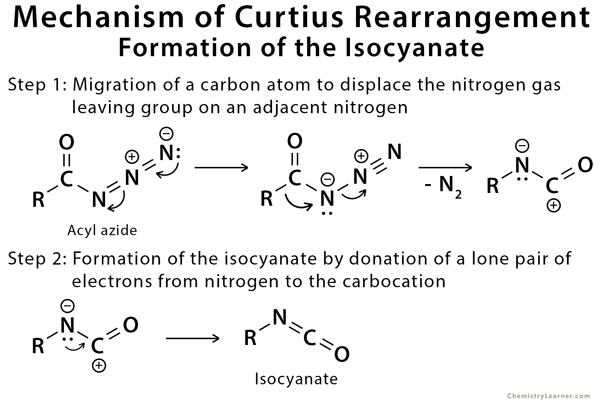
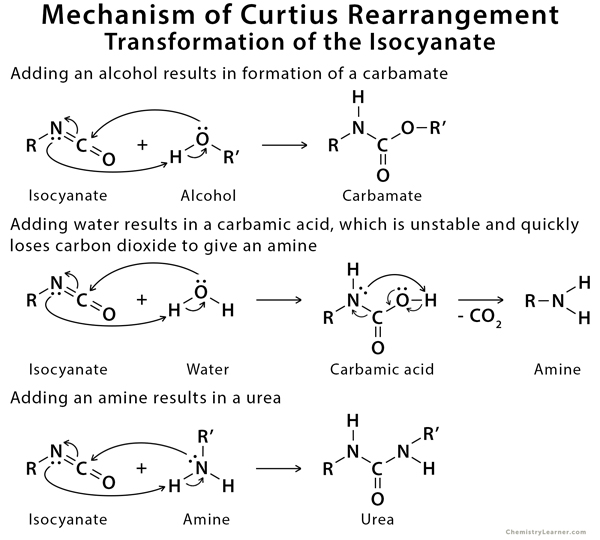
Mechanism of Curtius Rearrangement
The mechanism of Curtius rearrangement consists of a shift of the alkyl group from the carbonyl carbon to the closest nitrogen with the release of nitrogen gas. Subsequently, the isocyanate is formed and potentially reacts further in the presence of nucleophiles in solution to give rise to new stable compounds [3,4].
Applications of Curtius Rearrangement
There are several applications of the Curtius rearrangement in drug discovery and medicinal chemistry. Due to its tolerance for a large variety of functional groups and complete retention of stereochemistry during the rearrangement, the Curtius rearrangement has been utilized in the synthesis of a wide variety of medicinal agents with amines and amine‐derived functional groups such as ureas and urethanes [5].
References
- Definition – Ncbi.nlm.nih.gov
- Definition and example – Chemistryworld.com
- Definition and mechanism – Organic-chemistry.org
- Definition and mechanism – Name-reaction.com
- Applications – Researchgate.net