Dynamic Equilibrium
Table of Contents
What is Dynamic Equilibrium [1-4]
Equilibrium refers to the steady-state in a chemical reaction, where the reactant’s concentration is equal to the product’s concentration.
Dynamic equilibrium can be defined as the steady state of a reversible reaction occurring in a closed system. Both the forward and reverse reaction proceeds in the opposite direction with equal rates. The concentrations of reactants and products remain constant at equilibrium, provided the conditions (temperature, pressure, and composition) are not altered.
A reversible reaction achieves equilibrium when the forward and backward reactions occur at the same rate yielding both reactants and products. As a result, the amount of reactants converting into products and products transforming back into reactants remain constant. For this reason, this kind of chemical equilibrium is called ‘dynamic equilibrium’.
Characteristics of Dynamic Equilibrium
A few characteristics of dynamic equilibrium are listed below:
- The equilibrium can be achieved if the reversible reaction occurs in a closed vessel.
- At equilibrium, both the reactants and product are present.
- The forward and backward reactions move in the opposite direction with equal rates.
- The equilibrium state can be approached from either direction.
- The concentration of reactants and products, along with their temperature, pressure, and color, remains unchanged.
- A catalyst can accelerate the approach but does not affect the position of the equilibrium.
- According to Le Chatelier’s principle, a change in the observable properties of the system, such as temperature, pressure, or concentration, can shift the equilibrium point to the right or left side as required.
Examples of Dynamic Equilibrium [2,4]
Here are some examples of dynamic equilibrium:
- In a new bottle of an aerated drink, the carbon dioxide present in the liquid phase has a fixed value. When we pour half of the drink and reseal the bottle, the liquid CO2 slowly gets converted into gaseous CO2 to reach a new point of equilibrium. Here, the conversion rate of CO2 from gas to liquid and liquid to gas is equal.
- In the single-phase system, acetic acid undergoes dissociation, leading to a dynamic acid-base equilibrium.
CH3COOH ⇌ CH3COO– + H+
In the gaseous phase, the dimerization of nitrogen dioxide occurs: 2NO2 ⇌ N2O4
- Industrial synthesis of ammonia via Haber’s process
N2 (g) + 3H2 (g) ⇌ 2NH3 (g)
- If solid crystals of NaCl are added to its saturated aqueous solution, the crystals will simultaneously dissolve and recrystallize within the solution.
NaCl(s) ⇌ Na+(aq) + Cl–(aq)
The reaction will be in dynamic equilibrium when the dissolution rate of the NaCl equals the rate of recrystallization.
- Warm-blooded species, like humans, keeps the body temperature nearly constant by homeostasis. If the temperature goes up, our body cools down by sweating as the blood vessels near the skin’s surface dilate to allow heat to escape. On the other hand, if the temperature falls, the body shivers to create metabolic heat. The capillaries become smaller to retain heat and keep blood away from the arms and legs to maintain the core temperature.
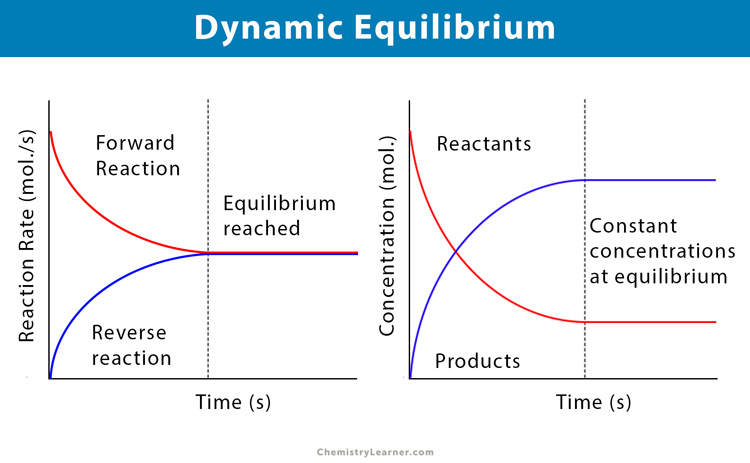
Dynamic Equilibrium Graph
The two graphs in the following diagram explain the state of dynamic equilibrium. The first graph, i.e., the reaction rate vs. time graph, depicts that the rate of both forward and reverse reactions becomes equal upon reaching the equilibrium point. On the other hand, the second one, i.e., the concentration vs. time graph, shows that the concentration of both reactants and products becomes constant at equilibrium.
Static and Dynamic Equilibrium [4]
Static equilibrium refers to a condition when the reaction occurring in the system comes to a halt. Therefore, the motion between reactants and the products ceases, leading to no exchange between reactants and products. Here, the equilibrium is attained once all the limiting reagents are used up.
Static vs. Dynamic Equilibrium
The key differences between static and dynamic equilibrium are as follows:
| Static Equilibrium | Dynamic Equilibrium |
|---|---|
| Both forward and backward reactions come to a halt. | The rates of forward and reverse reactions become equal. |
| Unidirectional (reactants to products) | Bidirectional (reactants to products & products to reactants) |
| No further changes happen in the mixture. | The changes continue to happen in the mixture to keep the composition equal. |
| Occurs in both open and closed system | Occurs in a closed system |
| Irreversible | Reversible |
FAQ
Ans. A change in body temperature is an example of dynamic equilibrium where balance is attained within an environment due to an internal control mechanism that continuously contrasts outside forces that tend to change that environment.