Epoxide Reactions
Epoxides are compounds that are very similar to ethers. They consist of a three-membered ring structure with an oxygen atom in one vertex and carbon atoms occupying the other two. Unlike ethers, epoxides are highly reactive. The reason is that the ring strain is relieved when a nucleophile attacks the electrophilic carbon atoms, resulting in a ring-opening reaction. Examples of epoxides include cyclohexene oxide and cyclopentene oxide [1-4].
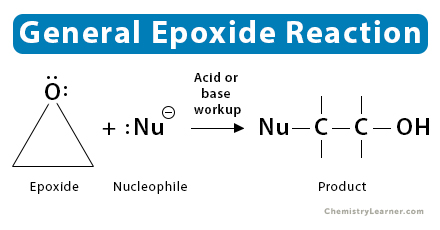
General Reaction
Types of Epoxide Reactions
Epoxides undergo nucleophilic substitution via the SN1 or SN2 mechanism, depending on the nature of the epoxide and reaction conditions. For an asymmetric epoxide, the product’s structure will depend on which mechanism dominates [1-4].
1. Alcoholysis
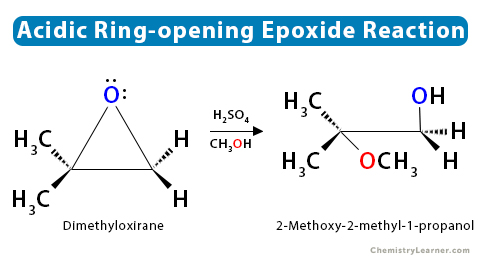
1.1 Acidic Condition
When an asymmetric epoxide undergoes alcoholysis in acidic methanol (CH3OH), the SN1 mechanism dominates, and the most substituted carbon is attacked.
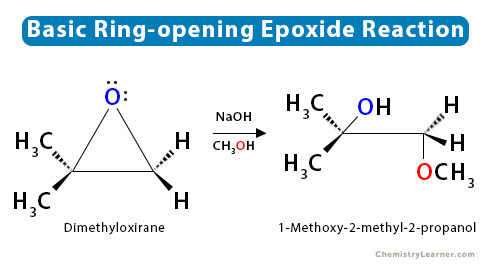
1.2 Basic Condition
When asymmetric epoxides undergo alcoholysis in basic methanol, the SN2 mechanism dominates, and the least substituted carbon is attacked.
For both the acidic ring-opening and basic ring-opening, the reactions are regioselective.
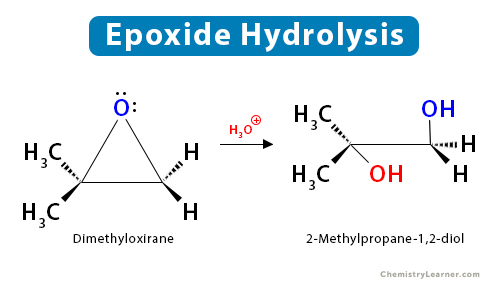
2. Hydrolysis
Epoxides are cleaved by hydrolysis to give trans-1,2-diols, also known as vicinal diols. They can be performed under acidic and basic conditions, resulting in the same regioselective reactions as in the case of alcoholysis.
2.1 Acid-Catalyzed Hydrolysis
When an asymmetric epoxide undergoes acidic hydrolysis, the incoming nucleophilic water molecule attacks the most substituted carbon through the SN2 mechanism.
2.2 Base-Catalyzed Hydrolysis
When an asymmetric epoxide undergoes basic hydrolysis, the incoming water nucleophile attacks the least substituted carbon through the SN2 mechanism.
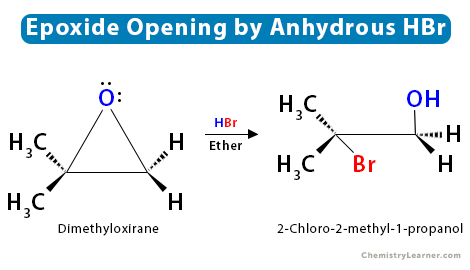
3. Reaction with Anhydrous Hydrohalic Acid (HX)
Epoxides can be opened by anhydrous hydrohalic acids (HX) to form a trans halohydrin. The halide will attack the least substituted carbon through the SN2 mechanism when both the epoxide carbons are either primary or secondary. However, if one of the epoxide carbons is tertiary, the halide will mainly attack the tertiary carbon through the SN1 mechanism.
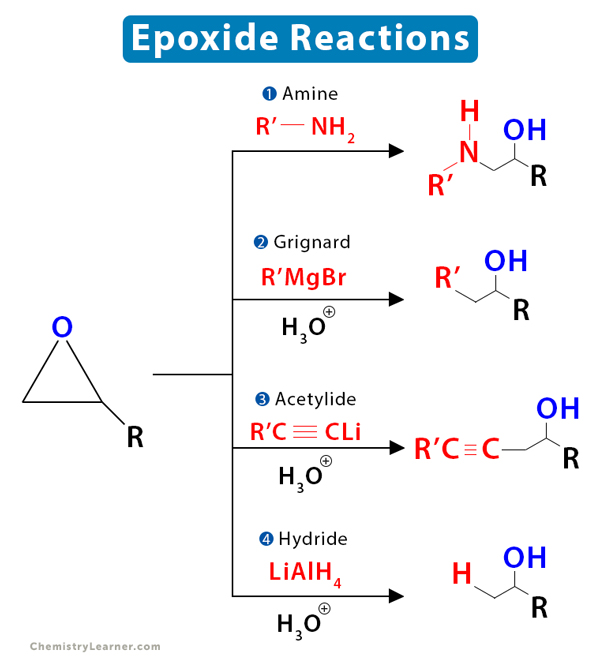
4. Reaction with Other Basic Nucleophiles
Basic nucleophiles, like Grignard reagent, can open an epoxide’s ring and form a wide range of products. Grignard reagents are both strong nucleophiles and strong bases. Hence, the reaction follows the SN2 mechanism. The result is a primary alcohol with two carbon atoms more than the original Grignard reagent.
The following image summarizes the reactions.
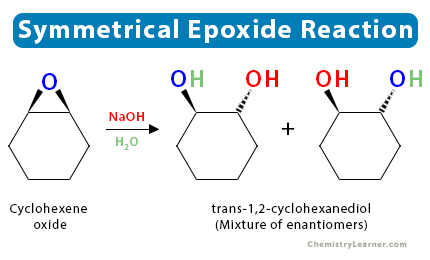
5. Symmetric Epoxide Reactions
In all the above reactions, we have considered asymmetric epoxides, where regioselectivity and stereochemistry of the reaction determine the product. However, if the epoxide is symmetrical, each carbon atom has the same probability of accepting the incoming nucleophile. In such a case, the products formed are a mixture of enantiomers.
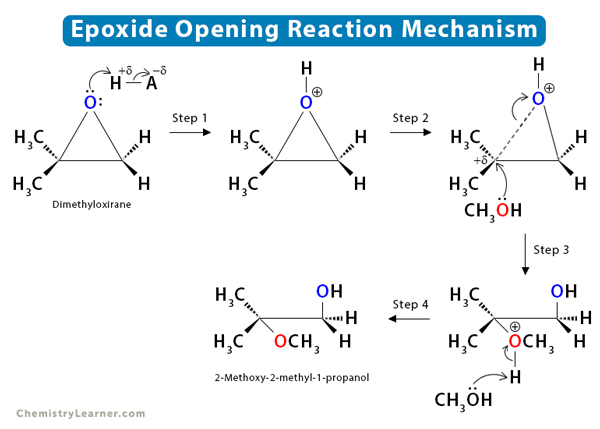
Mechanism of Epoxide Ring-opening
To understand the mechanism of the epoxide ring-opening reaction, let us look at the acid-catalyzed alcoholysis [1-4].
Step 1: The acid (HA) protonates the oxygen, resulting in a good leaving group.
Step 2: The carbon-oxygen bond breaks, and a positive charge develops on the most substituted carbon.
Step 3: The nucleophile attacks the least hindered electrophilic carbon, producing a regioselective product. The attack occurs from the back side, as in an SN2 reaction.
FAQ
Ans. Lithium aluminum hydride (LiAlH4) reduces an epoxide to produce the corresponding alcohol.





Am so satisfied with this ????