Home / Organic Chemistry / Grignard Reaction
Grignard Reaction
Table Of Contents
Definition: What is Grignard Reaction?
Grignard reaction is a type of organometallic chemical reaction in which a Grignard reagent is added to water, alcohol, aldehyde, ketone, ester, and carbon dioxide. The resulting product is a compound consisting of alkane or alcohol [1].
Mechanism of Grignard Reaction
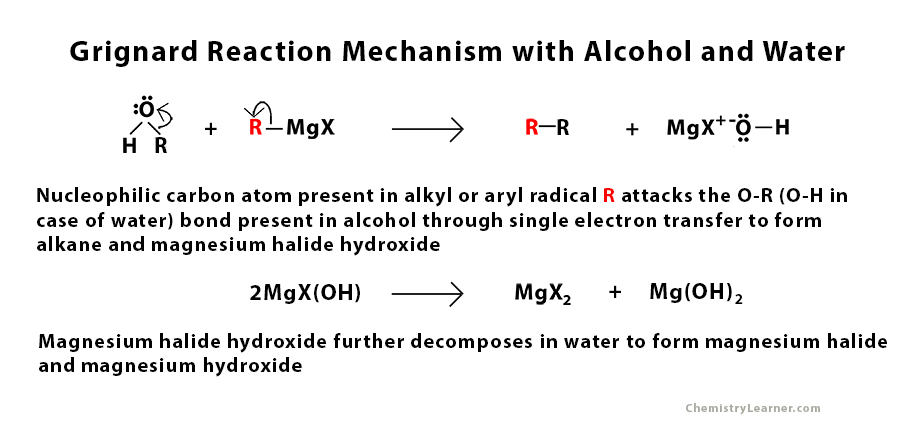
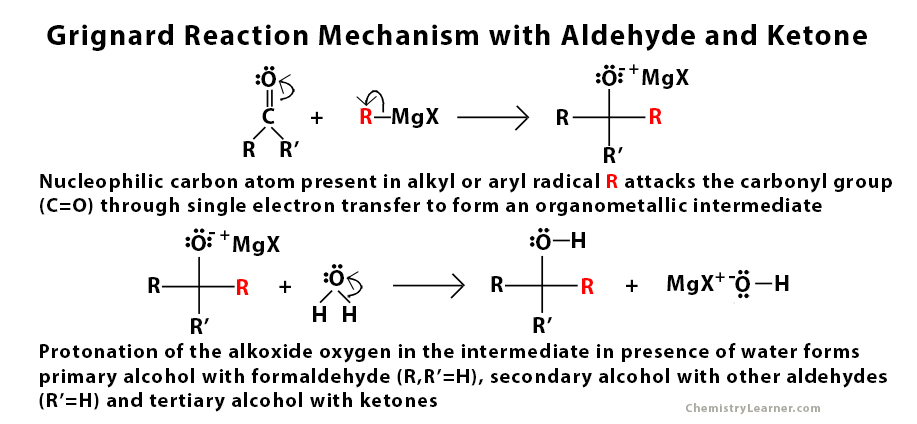
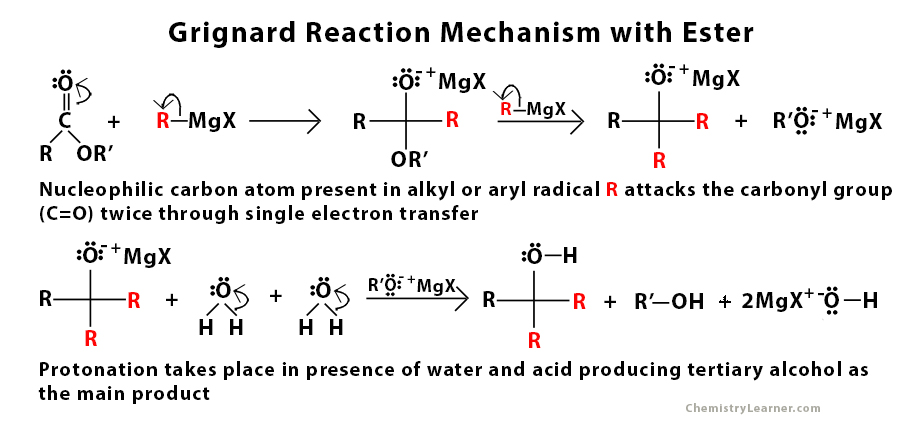
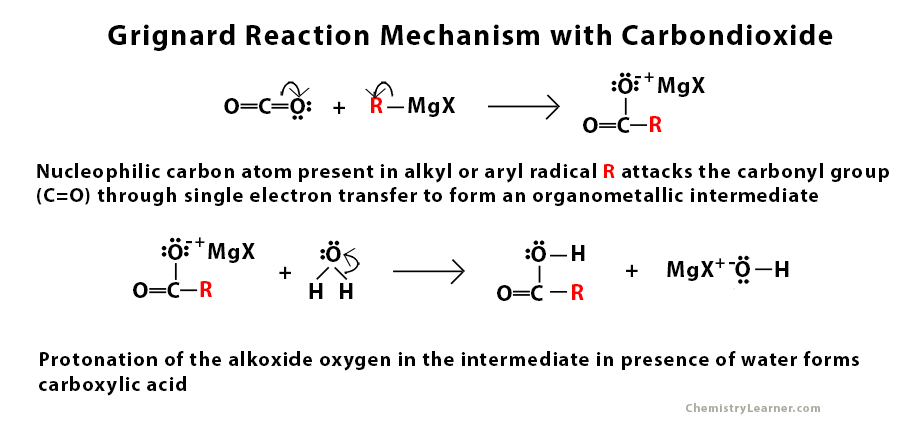
The Grignard reagent is very reactive due to the nucleophilic nature of the carbon atom present in the alkyl or aryl radical. It is used to perform a nucleophilic addition reaction. The main role of this nucleophile is to attack the electrophilic carbon atom that is present in the carbonyl group that is present in aldehyde, ketone, and ester through a single electron transfer mechanism [2]. This step is followed by protonation in the presence of water and acid.
Grignard Reaction Mechanism with Water and Alcohol
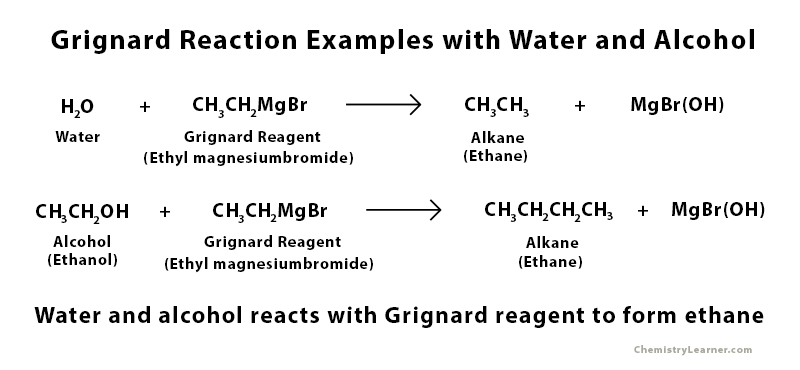
Examples of Grignard Reaction with Water and Alcohol
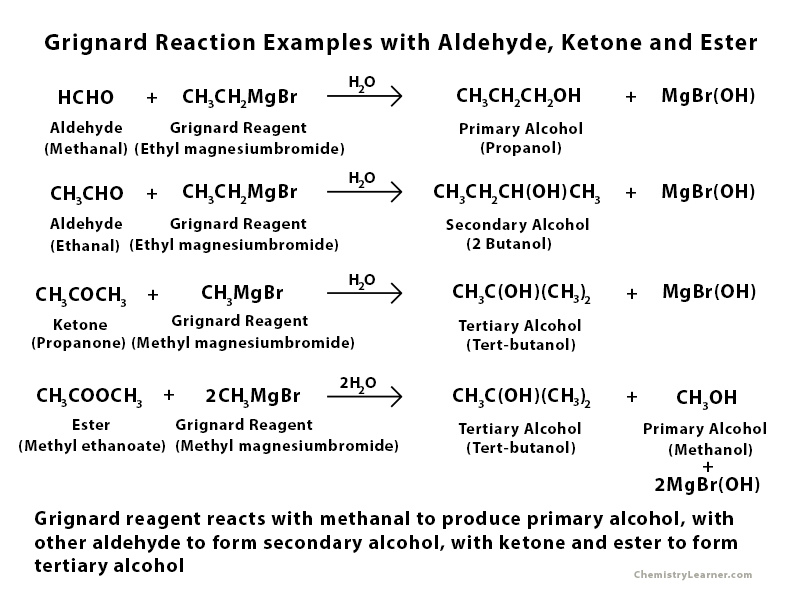
Grignard Reaction Mechanism with Aldehyde and Ketone
Grignard Reaction Mechanism with Ester
Examples of Grignard Reaction with Aldehyde, Ester, and Ketone
Grignard Reaction Mechanism with Carbon dioxide
Examples of Grignard Reaction with Carbon dioxide
- References
- Definition of Grignard reaction – Organic-chemistry.org
- Mechanism of Grignard reaction – Study.com
- Mechanism and examples of Grignard reaction – Chemguide.co.uk
- Grignard reaction of alcohol – Socratic.org
- Grignard reaction with ester –Chemistryscore.com
- Grignard reaction with ester – Chem.libretexts.org
- Grignard reaction with ester – Chem.ucalgary.ca






Please give reaction of grignard reagent with ethylamine