Haloform Reaction
Definition: What is Haloform Reaction?
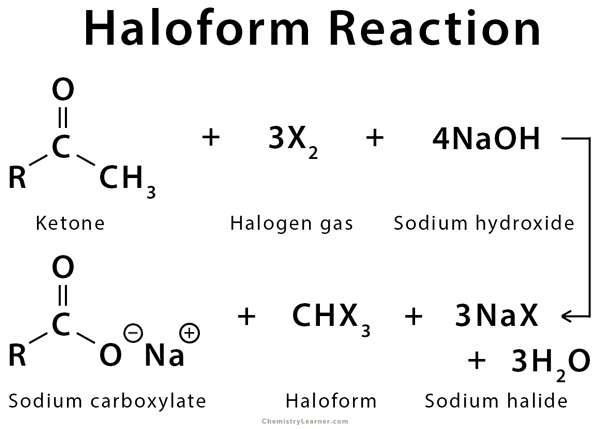
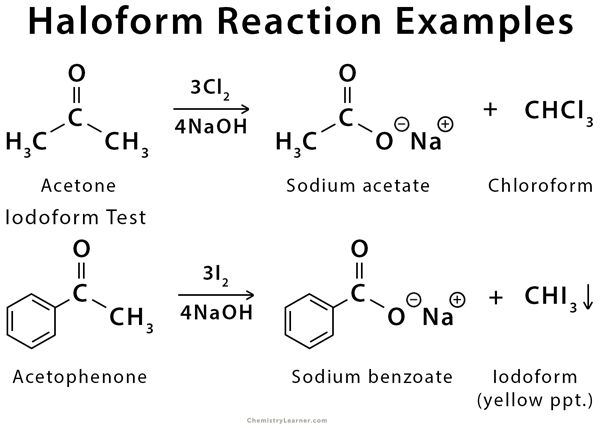
The Haloform reaction is a chemical reaction where haloforms (CH3X) are produced by the complete halogenation of acetone (CH3COCH3), acetophenone (PhCOCH3), or acetaldehyde (HCOCH3) in the presence of a base. The aldehyde or ketone must have a methyl group next to the carbonyl since methyl has three alpha-hydrogen atoms. The reaction can be used to transform acetyl groups into carboxyl groups or to produce chloroform (CHCl3), bromoform (CHBr3), or iodoform (CHI3) and also cyanide. This reaction has been used in qualitative analysis to indicate the presence of a methyl ketone in which excess iodine is used to halogenate the compound. The product iodoform precipitates as a yellow-colored substance and has a characteristic odor [1-5].
The history of the haloform reaction can be traced back to 1822 when French professor Georges-Simon Serullas made the accidental discovery that iodine and alkali convert alcohol into iodoform.
Examples of Haloform Reaction
A typical example of this reaction is the conversion of acetophenone into benzoic acid [1-4].
Mechanism of Haloform Reaction
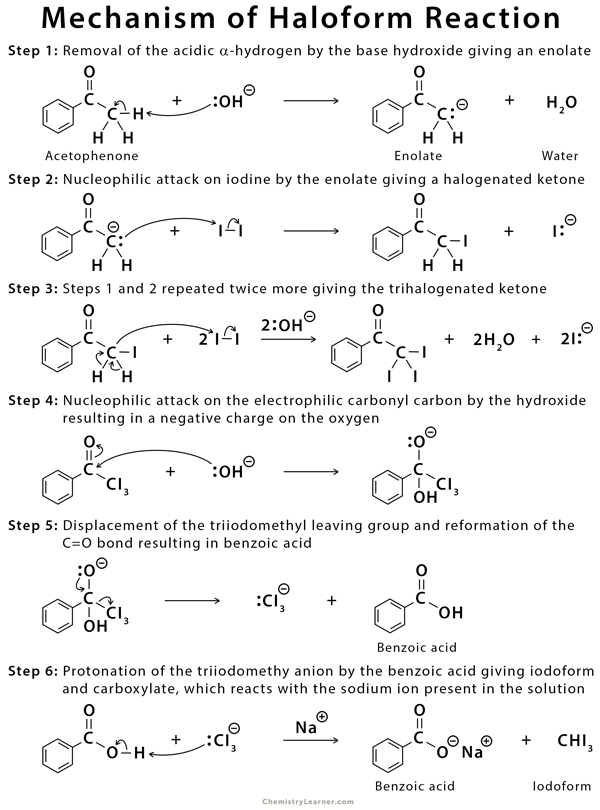
When methyl ketones are treated with the halogen in basic solution, the substitution of the three alpha-hydrogens in the methyl group halogen atoms takes place (polyhalogenation). The hydrogens replaced by halogens are all from the same side of the carbonyl group, especially in symmetric compounds like acetone (CH3COCH3). Once hydrogen is replaced by halogen, the other hydrogen atoms at that carbon become more acidic, causing further substitution at the same place. This step is followed by the cleavage of the substituted methyl group [1-6].
References
- Definition, example, and mechanism – Chem.libretexts.org
- Definition, example, and mechanism – Chem.ucalgary.ca
- Definition, example, and mechanism – Organic-chemistry.org
- Definition, example, and mechanism – Pubs.acs.org
- Definition and mechanism – Sciencedirect.com
- Mechanism – Leah4sci.com