Halogenation
What is Halogenation (1)
Halogenation is a chemical reaction in which a halogen is added to a hydrocarbon by replacing one of its hydrogens. The product thus formed is known as a halogenated compound. It occurs in both saturated and unsaturated hydrocarbons and aromatic hydrocarbons.
The most common halogens in this reaction are fluorine, chlorine, bromine, and iodine. The order of reactivity for these halogens is fluorine (F) > chlorine (Cl) > bromine (Br) > iodine (I). F is the most aggressive and produces the most stable halogenated compound. Thus, the electronegativity of the halogen atom is a driving force for these reactions.
Halogenation reactions are essential in producing several products, such as pharmaceutical goods, refrigerants, polymers, fuel additives, fire retardants, etc.
Types and Examples of Halogenation (2-7)
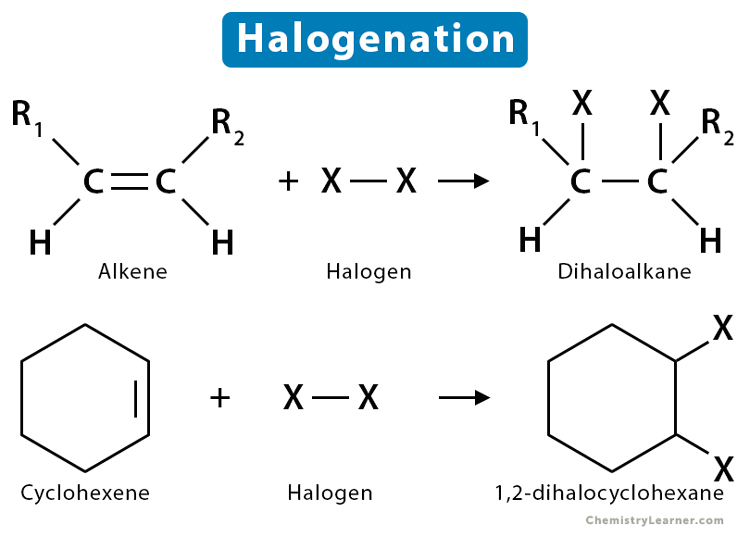
1. Halogen Addition Reaction
As the name suggests, an atom of halogen is added to a hydrocarbon. This reaction is common in unsaturated carbons, alkenes, and alkynes.
1.1 Halogenation of Alkene
It is the addition of a halogen to the C=C double bond of an alkene. The alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as Cl2 or Br2 is added to a molecule after breaking the carbon-to-carbon double bond. The halides add to neighboring carbons from opposite faces of the molecule. The resulting product is a vicinal (neighboring) dihalide, where the halogens are attached to adjacent carbon atoms.
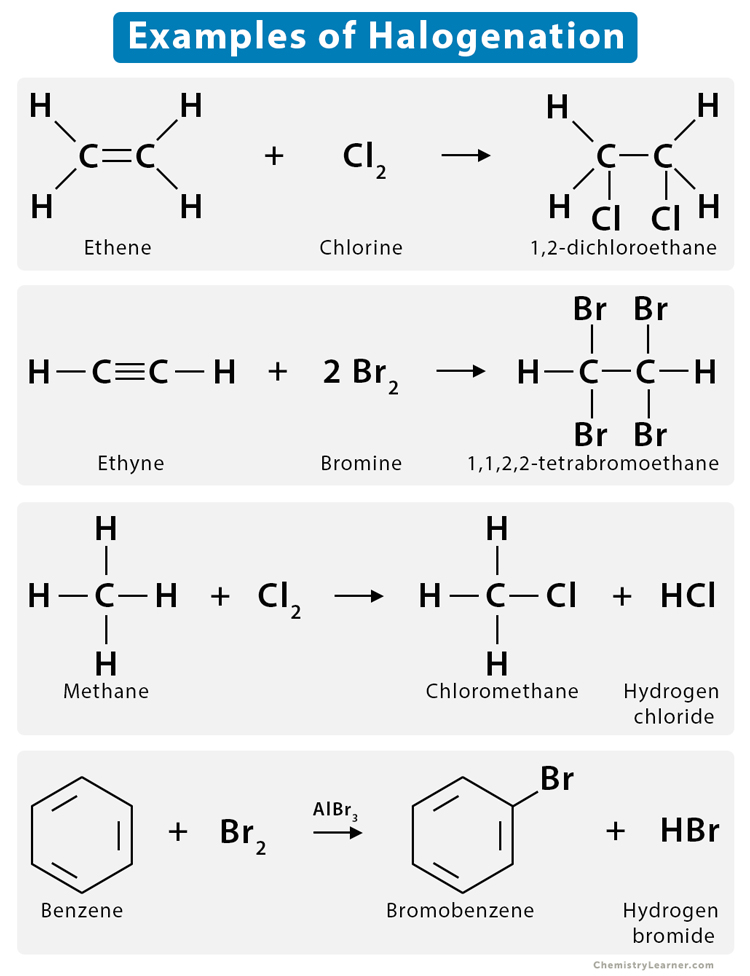
Example: Reaction of ethene (C2H4) with chlorine (Cl2)
Ethene is a symmetrical alkene containing two carbon atoms. It reacts with chlorine in the presence of carbon tetrachloride (CCl4), producing dichloroethane (CH2Cl – ClCH2).
CH2 = CH2 + Cl2 → CH2Cl – ClCH2
1.2 Halogenation of Alkyne
Alkynes react with halogens in the same way as alkenes. Adding the halogen to the π-bond produces a trans or vicinal dihalide, which is then converted into a tetrahalide.
Example: Reaction of acetylene (CH ≡ CH) with bromine (Br2)
Acetylene reacts with bromine in the presence of CCl4, producing 1,1,2,2-tetrabromoethane (HBr2C–CBr2H)
Step 1: CH ≡ CH + Br2 → HBrC = CBrH
Step 2: HBrC = CBrH + Br2 → HBr2C–CBr2H
2. Halogen Substitution Reaction
In a substitution reaction, one atom is swapped with another atom.
2.1 Halogenation of Alkane
Alkanes undergo a substitution reaction with halogens in the presence of light.
Example: Reaction of methane (CH4) with bromine (Br2)
Methane reacts with bromine in the presence of ultraviolet light, forming bromomethane (CH3Br) and hydrobromic acid (HBr). Here, one of the hydrogen atoms from the methane is replaced by a bromine atom.
CH4 + Br2 → CH3Br + HBr
2.2 Electrophilic Aromatic Substitution
Unlike aliphatic compounds, aromatic compounds like benzene do not undergo addition reactions. In this case, the addition of electrophiles, like Cl2 and Br2, results in substitution.
Example: Reaction of benzene (C6H6) with bromine (Br2)
Benzene reacts with bromine in the presence of ferric bromide (FeBr3), producing an aryl halide, bromobenzene, along with hydrobromic acid (HBr). Here, one hydrogen atom of benzene gets substituted by bromine.
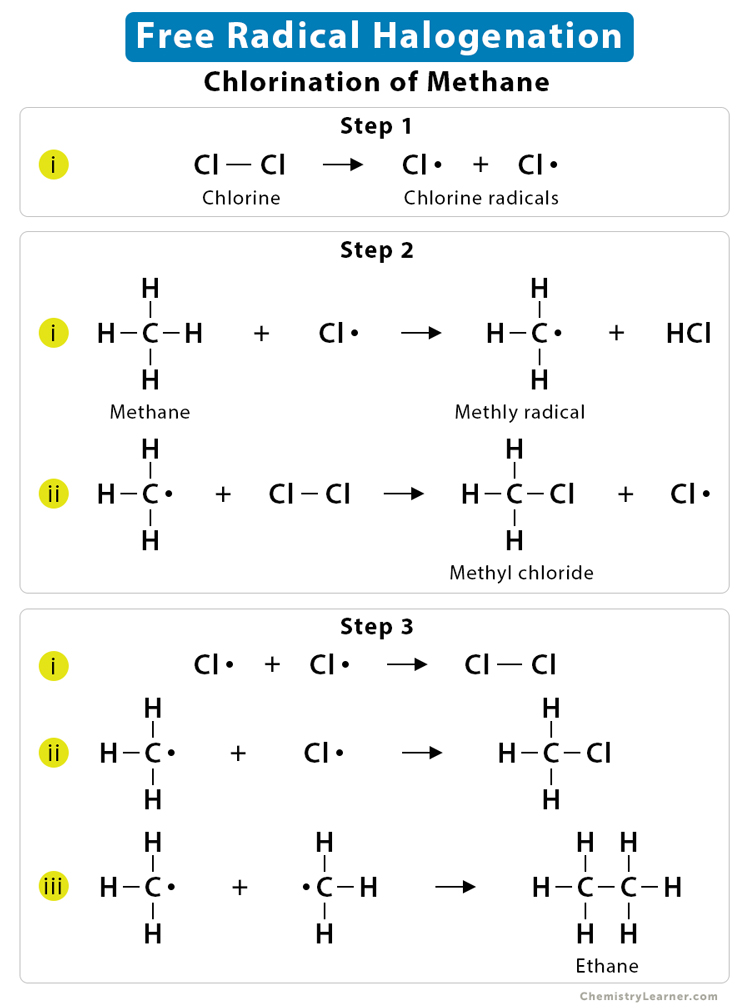
3. Free Radical Halogenation
Free radical halogenation is a photochemical reaction that substitutes chlorine or bromine for hydrogen on an alkane.
These reactions occur in 3 steps, which are as follows:
Step 1: Initiation
In this step, the halogen splits into its two free radicals (Cl•) in the presence of UV light.
Cl – Cl → Cl• + Cl•
Step 2: Propagation
Next, a hydrogen atom is pulled off from the subjected hydrocarbon.
Here, let us take the example of methane (CH4). A hydrogen atom is removed from methane during this stage, thus producing a primary methyl radical. The methyl radical then pulls a Cl• from Cl2.
CH4 + Cl• → C•H3 + HCl
C•H3 + Cl – Cl → CH3Cl + Cl•
Step 3: Termination
In this step, the two free radicals recombine to destruct the free-radical intermediates.
Cl• + Cl• → Cl – Cl
C•H3 + Cl• → CH3Cl
C•H3 + C•H3 → CH3– CH3
∴ The net reaction is:
CH4 + Cl2 → CH3Cl + HCl
4. Ketone Halogenation
It is a specific halogenation reaction, carried out either under acidic or basic conditions. Here, a halogen can be installed selectively in the alpha position of a ketone. It can be readily done due to the ability of ketone to form an enolate in basic solution or an enol in acidic solution.
Example: Bromination of acetone
Bromination of acetone (CH3-CO-CH3), carried out under either acidic or basic conditions, to give bromoacetone (CH3COCH2Br). It is an alpha halogenation of ketone.
CH3-CO-CH3 + Br2 → CH3COCH2Br + HBr
Mechanism of Halogenation (2-3)
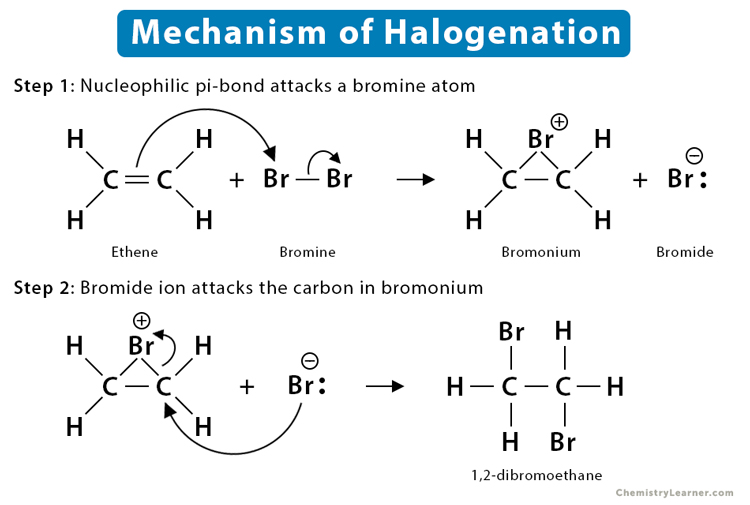
1. Halogen Addition Reaction
In the alkene halogenation reaction, a dihalide such as Cl2 or Br2 gets added to a molecule after breaking the double bond between carbon atoms. The halides add to neighboring carbons from opposite faces of the molecule. The resulting product is a vicinal (neighboring) dihalide.
The overall reaction takes place in the following steps:
- The nucleophilic pi bond of the alkene attacks a halogen. The pi bond breaks in the process. The pi electrons will then reach out to grab the temporarily partially positive halogen.
- As soon as the halogen is attacked, it will lose its partial positivity. Next, the halogen attacks back, using one of its lone electron pairs. It results in the formation of a halogen bridge. The bridged intermediate is called halonium – for instance, bromonium or chloronium, when bromine or chlorine is involved.The unaffected halide grabs the bonding electrons and breaks away from the molecule.
- Bridged halogen has a charge of +1 due to 2 bonds and 2 lone pairs.
- Despite having a complete octet, the negative charge of the second halide makes it highly nucleophilic. As a result, it gets attracted to partially positive carbon atoms in the bridged molecule.
- So, the second halide returns and attacks from the opposite side of the molecule, breaking the bridge.
- The resulting product is called a vicinal dihalide, having 2 halogens attached on opposite sides or ‘anti’ to each other.
Example: Reaction of 2-butene (CH3CH=CHCH3) with chlorine (Cl2)
2-butene reacts with chlorine, in the presence of CCl4, forming 3-chloro-2, 2-dimethylbutane [CH3-CH(Cl)-CH(Cl)-CH3].
CH3CH=CHCH3 + Cl2 → CH3-CH(Cl)-CH(Cl)-CH3
2. Halogen Substitution Reaction
Alkanes undergo a substitution reaction with halogens in the presence of light. It is a simple substitution reaction in which a C-H bond is broken, forming a new bond between carbon and halide.
The whole reaction occurs in 2 steps. The process of alkane halogenation is described below using the reaction chlorination of methane (CH4).
Step 1: In the first step, the covalent bonds between the chlorine atoms are broken, forming free radicals of chlorine.
Cl – Cl → Cl• + Cl•
Step 2: In the next step, one free radical of chlorine attacks the bond between C-H. Then it replaces a hydrogen atom of methane. As a result, chloromethane (CH3Cl) is formed. The released hydrogen gas binds with the remaining free radical of chlorine, forming hydrochloric acid (HCl).
CH4 + Cl• → CH3Cl + ½ H2
½ H2 + Cl• → HCl
References
- Addition of Halogens – Chem.libretexts.org
- The Halogenation Of Alkenes – Chemguide.co.uk
- Halogenation of Alkanes – Chem.libretexts.org
- Electrophilic Addition of Halogens to Alkenes – Chem.libretexts.org
- Alkyne Halogenation: Bromination, Chlorination, and Iodination of Alkynes – Masterorganicchemistry.com
- Free Radical Halogenation of Alkanes – Chem.libretexts.org
- Alpha Halogenation of Aldehydes and Ketones – Chem.libretexts.org